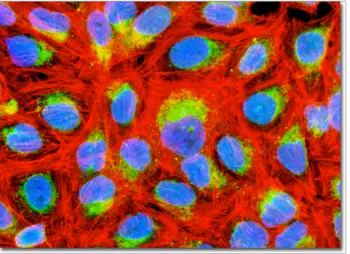
Immunofluorescence in Microscopy
Applications, Direct and Indirect Methods
Immunofluorescence is a common technique using a fluorescence microscope in labs/institutions that perform biological studies, as it allows scientists to easily identify and differentiate between the antibodies and antigens present in a tissue sample.
This method of study focuses on the immune response that occurs within a diseased tissue or its cells, a behavior researchers can observe after they apply a fluorescent stain either directly or indirectly to the specimen.
In studying these cells and their reactions, scientists are able to determine which specific proteins protect the body against those particular foreigners that invade it with the intent of causing sickness or in some cases, death.
Applications
Scientists can use the immunofluorescence technique to perform a variety of lab tests and observations, each of which provides a fresh perspective for the sample currently undergoing analysis.
For instance, a researcher may opt to use this testing method to examine tissue samples and beads, detect particular proteins using microarrays, or they might choose to evaluate cultured cells, as well as those in suspension.
An observer can apply this technique to samples that are fixed or fresh, thus providing more opportunity for diverse analysis and accurate results.
A researcher may also choose to apply this technique when studying DNA sequences on chromosomes, as these particles are extremely small and hard to detect. Obtaining spatial data concerning tissue or cell genetics is another way to use immunofluorescence, as is the observation of parasites and bacteria.
The deeper layers of a cell or tissue, along with the antigens that infect these layers, is another area that scientists can study using this staining method.
- Global leader in development of products for biotechnical research -
Purpose
The reason scientists use immunofluorescence in lab studies is because it uses fluorescent dyes that attract and bind to specific antibodies made by the immune system when it is under attack by a particular antigen.
The newly made antibody attaches to the antigen, either directly or indirectly, allowing scientists to observe the foreign invader, its behavior and the reaction given by the antibody.
A researcher can then use a confocal or fluorescence microscope to study the now colorful antibody-antigen pair and they can also obtain a count of the specimens in their sample by using other tools such as an array scanner, flow cytometer or an automated imaging instrument.
Direct Approach
Researchers tend to use direct immunofluorescence less often than the indirect method because the former involves the direct staining of the specific antibody that attaches to the specific antigen that the scientist wants to observe.
It also is a less advantageous method, as it provides a high cost, little flexibility and a poor signal. On the plus side, this technique does offer timesaving advantages, such as simplicity in the labeling processes and less waiting when applying and drying stains.
Indirectly
It is not too often that an indirect approach is the better choice, but when it comes to immunofluorescence, it is the clear winner.
A complex method that requires a secondary antibody, known as an anti-immunoglobulin antibody, receives the fluorescent dye as opposed to the primary antibody, or the one that binds to the particular antigen of interest.
This process is highly sensitive but produces better results than the direct staining method, as it enhances the image observed by the scientist.
By staining the secondary antibody, many of which can attach to a single primary antibody, the observer can see a fluorescent image of the specimen that looks more like a firework display than a faint flickering of a single candle.
In addition to providing a top-notch image, indirect staining also tends to be less expensive than the direct method, offers better quality control and the researcher can purchase the antibodies in a variety of colors.
However, like anything else, this method does have its disadvantages, such as incompatibility or a poor reaction between the secondary and primary antibodies or the antigen.
Tests requiring multiple samples and labels often become laborious and time-consuming, as the researcher may need to acquire different isotopes or primary antibodies raised from a different species to help avoid cross-reactions and obtain the most accurate results.
Staining
The type of stain you use, known generally as a fluorochrome, and whether or not you apply a mordant or deionizer, all depends on the specimen undergoing the analysis.
In immunofluorescence, your stain is always going to be one that is fluorescent, like tetramethyl rhodamine isothiocyanate (TRITC) or fluorescein isothiocyanate (FITC).
For high levels of fluorescence, a researcher may opt to adhere the sample to the slide with gluteraldehyde, though making a sample too bright is not always good.
To reduce the level of brightness, the scientist can, prior to incubation, rinse the sample with 0.1 percent of sodium borohydride in a phosphate-buffered saline.
Another problem scientists can run into when preparing samples with immunofluorescence stains is that they can overlap, thus making it hard to read the results or providing false data. To correct this problem, the scientist needs to electronically remove the dyed specimens that crossed-over using computerized tools.
Photobleaching, or the destruction of the fluorochrome, can prove to be another obstacle when conducting immunofluorescence, as the loss of color, and even degradation in part or in whole of the specimen, forces the scientist to re-start the entire experiment.
To obtain the best results, a scientist should try to use a fluorochrome that provides a bright image and comes out clear in photos.
It should also be one that a fluorescence detection instrument can measure when the absorption levels reach their peak and it should be resilient to alterations that can occur within the test environment or when adhering to the antibody.
Immunofluorescence is not the most complex of microbiological methods, but it is still very useful when conducting a variety of studies and it is a good point at which to start.
By simply, and accurately, adding the correct dye to the right protein, the specimen undergoing analysis lights up like a star in the night sky, giving the observer a crystal clear view of the sample's occupants and activities.
The ability to differentiate between those proteins that protect the body and those that seek to destroy it is crucial to creating new medicines, developing treatment plans and saving lives.
Related Articles: Understanding the Green Fluorescent Protein, Fluorescent Dyes and Total Internal Reflection - Definitions, Applications and Uses in Microscopy (TIRF)
More on Histochemistry - Techniques/Methods, Staining and Tests
Return to Widefield Epifluorescence Microscopy
Return from Immunofluorescence to Fluorescence Microscope
Return to Best Microscope Reviews and Microscopy Research
Find out how to advertise on MicroscopeMaster!