Histochemistry
Techniques/Methods, Staining and Tests
Introduction
Histochemistry is an important technique that is used for the visualization of biological structures. As such, it is concerned with the identification and distribution of various chemical components of tissues through the use of stains, indicators as well as microscopy.
Essentially, identification and distribution of chemical constituents of tissues is achieved through the exploitation of unique chemical environments in cells, heterologous expression techniques as well as enzymatic activities.
Histochemical Techniques/Methods
Perl's Reaction
This method is particularly important for the detection of ion levels (ferric ions). Because it can help detect the presence of ferric ions, this technique is used to determine the level of these ions in such organs as the spleen and bone marrow. It can be used to tell whether there are excessive amounts of the ion as observed in hemochromatosis (excessive levels of ferric ions with deposits in the liver and pancreas) or hemosiderosis where deposits can be found in the liver, spleen and the lymph nodes.
In plants, this technique has also been shown to help understand the homeostasis of ferric ions.
Principle
With this technique, ferric ions present in the tissue will combine with ferrocyanide resulting in the formation of a pigment called Prussian blue (ferric ferrocyanide). In plants, the technique is also based on the conversion of ferrocyanide into insoluble crystals (Prussian blue) in the presence of Ferric ions under acidic conditions. Prussian blue (resulting from the reaction) is bright blue in color, which indicates the presence of ferric ions.
Staining procedure
In Perl's reaction technique, a known positive control tissue is used as control while 10 percent formalin is used as a fixative.
Requirements:
- Microwave oven
- Acid cleaned glassware
- Non-metallic forceps
- 5 percent potassium ferrocyanide
- 5 percent hydrochloric acid
- Sample section (cut to about 4µ)
* It's important to use gloves, goggles and a lab coat because some of the chemicals used for this procedure can cause irritation.
Procedure
- Deparaffinize the section(s) and hydrate with distilled water
- Microwave for about 30 seconds and allow the specimen to stand in the working solution for about 5 minutes in the fume hood. The section can be treated with the working solution (acid ferrocyanide) for between 10 and 30 minutes.
- Rinse/wash the section using distilled water
- Stain (lightly) the section with 0.5% aqueous neutral red or 0.1% nuclear fast red. This part of the procedure is used to stain the nuclei.
- Rapidly wash the section using distilled water
- Dehydrate the section, clear and mount on the microscope stage for viewing
When viewed under the microscope, blue parts are indicative of iron while the red and pink parts indicate the nuclei and background respectively.
Von Kossa Technique
This is also an ion based technique that is used in histochemistry. It is a more sensitive technique that can be used to identify the presence of calcium deposits on cyst fluids, ductal ectasia and papillomatosis. However, excessive amount of calcium may be found in any given part of the body and can be demonstrated using the Von Kossa technique.
Although this technique is used to demonstrate the presence of calcium, it demonstrates an anion rather than the calcium ion itself.
Principle
For this technique, the sample section is treated with the solution of silver nitrate and is reduced and the calcium (if present in the sample) is reduced by the strong light and replaced with deposits of silver. As a result, it's visualized as metallic silver.
Requirements for Von Kossa technique
- A control sample (a tissue that contains known calcium deposits or undecalcified bone)
- 10 percent formalin (fixative)
- Glassware (clean)
- 60 watt lamp
- A mirror or foil
- 5 percent of silver nitrate solution
- 5 percent hypo (sodium thiosulfate)
Procedure
- Deparaffinize and hydrate the section using distilled water
- Place the section in the silver solution (in a glass jar) and place it in bright light (or in front of the 60 watt lamp). Place a mirror or a paper foil behind the jar so as to reflect the light. Leave it standing for about one hour or until the calcium turns black
- Rinse the section in distilled water
- Stain using 5 percent hypo solution for about 5 minutes
- Wash the section using tap water or rinse in distilled water
- Introduce the sample to nuclear-fast Red for about 5 minutes
- Wash using distilled water
- Dehydrate and mount for viewing
Observation (results)
A black color indicates the present of calcium (calcium salts), red indicates the nuclei while the cytoplasm will appear pink.
Lipids Staining
This technique is dependent on dyes that are soluble in lipids.
Some of the most common dyes used include:
- Sudan VI
- Sudan black
- Oil Red O
- Nile blue
Lipid staining is a useful technique that is used for demonstrating intracellular lipids in various tissue sections.
Principle
For this technique, the dye is more soluble in the lipid, which allows it to be more demonstrated than in the vehicular solvent. The dyes used in this technique are all interchangeable, which means that they can be substituted for each other for the staining process.
Staining procedure
Requirements/reagents
- ORO (Oil Red O) solution
- Glycerine jelly mounting medium
Procedure
- Cut the sample to obtain sections of between 8 and 10 microns and air dry
- Rinse the section with 60 percent isopropanol
- Stain the section with the Oil Red O working solution for about 15 minutes
- Rinse the specimen with 60 percent isopropanol
- Dip the section in Alum hematoxylin a few times in order to stain the nuclei
- Rinse in distilled water
- Mount the specimen in water or in glycerin jelly
Observation
Red color indicates the lipid while blue coloration indicates the nuclei.
Lipid staining technique is useful for showing the normal distribution of lipids as well as disease-related lipid accumulation.
Protein and Amino Acids
Some of the methods used for specific amino acids include:
- Millon's reaction
- Sakaguchi reaction
- Tetrazotized benzidine reaction
Millon's Reagent
Millon's reagent is used for detecting amino acid tyrosine.
Principle
In this technique, the mercurous and mercuric nitrate (components of the reagent) reacts with hydroxybenzene radicals to form a compound that is red in color. Tyrosine contains the phenolic group, which forms the red coloration in the presence of Millon's reagent. The compound formed through this reaction is called mercuric fumarate.
Milton's method procedure:
- Add about 2ml of the protein solution into a test-tube
- Add a few drops of the reagent (Millon's reagent) in to the test-tube that contains the original solution
- Using a tube holder, hold the test-tube over boiling water or flame and boil for about half a minute.
Observation
If the solution turns reddish in color after boiling, then tyrosine is present in the solution.
Sakaguchi Test
The Sakaguchi reaction test involves the use of the Sakaguchi reagent. This reagent is composed of 1-naphthol and sodium hypobromite and forms a reddish compound when mixed with the sample containing a Arginine.
The test is positive for any amino acid that contains the guanidine group in Arginine. Therefore, the Guanidine group in an amino acid will react with the α-Naphthol and alkaline hypobromite in the reagent to give of a red-colored complex indicating the presence of such amino-acids.
Sakaguchi's test procedure:
- Add 1 ml of the protein solution in to a test-tube
- Add 2 or 3 drops of 40 percent of sodium hydroxide, 2 drops of ethanolic a-Naphthol and 5 drops of bromide water
- Mix the contents in the test-tube well
- If a red-color complex forms when shaking the contents, then it is positive that the solution contains Arginine or a protein containing Arginine.
Tetrazotized Benzidine Reaction
Although it has been shown to be less effective, Tetrazotized benzidine is used in histochemistry to detect non-collagen proteins. The procedure involves the coupling of Tetrazotized benzidine with beta naphthol or Hyaluronic acid.
The method, commonly referred to as Tetrazotized benzidine helps in the detection of such non-collagen proteins as
tyrosine, histidine as well as tryptophan.
Nucleic Acids and DNA
Feulgen's Reaction
This is a relatively new technique that is used for demonstrating DNA in tissue sections. It is a sensitive means of detecting aldehydes, which makes it the ideal method for detecting the presence of DNA. Here, the section is treated with dilute hydrochloric acid in order to remove the bases.
The sugar part that remains reacts as an aldehyde ultimately forming a visible color.
Therefore, this method can be said to be divided in to two main parts:
1/ The first part of the procedure is the hydrolysis phase that involves the use of 5N HCl, ambient temperature for 40 minutes. This step is aimed at separately selecting 2 purine bases (adenine and guanine) which are removed from the DNA molecule.
2/ The second step is the staining phase. The reagent used is preferred because it is highly selective for DNA rather than RNA. Here, RNA does not react because of the presence of hydroxyl on carbon 2 of ribose, which prevents the acid (HCl) from hydrolyzing sugar. The reaction is also precise for the localization of DNA given that deoxyribose radicals are bound to phosphoric acid of the apurinic acid molecule following the removal of purine bases.
Some of the stains used for both DNA and RNA include:
- Methyl green pyronin stain
- Acridine orange
Saccharides
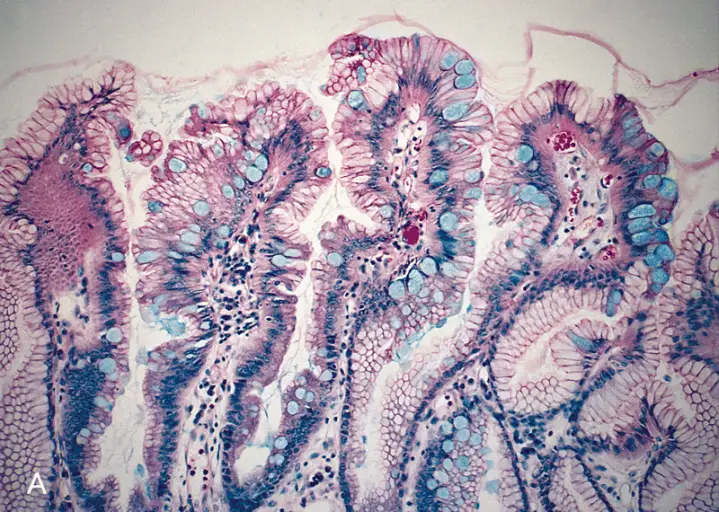
PAS reaction (Periodic Acid Schiff)
This is one of the most popular histochemical techniques for the detection of glycogen. It has been shown to be one of the best techniques for demonstrating carbohydrates in tissue.
In this technique, the periodic acid oxidizes tissue carbohydrates to produce aldehyde groups. This group then condenses with the reagent to form a bright red coloration to demonstrate the tissue component with carbohydrate attachments.
The diastase and a-amylase in the reagent act on the glycogen and depolymerize it into smaller sugar units (maltose and glucose) which are then washed out of the section.
Procedure
- Deparaffinize and hydrate the slide using distilled water
- Place the section in preheated diastate solution (at 37 degrees centigrade) for about an hour
- Wash the sample in running water for about 5 minutes
- Place the sections in 0.5 percent periodic acid solution for about 5 minutes
- Wash the section in distilled water
- Place the section in Schiff reagent for about 15 minutes
- Wash the section for about a minute in 0.55 percent potassium metabisulfite in order to remove excess stains
- wash in running tap water for about 10 minutes
- Counterstain using Harris's hematoxylin with acetic acid for half a minute
- Wash with running water
- Dehydrate with two changes using absolute alcohol, clear with xylene and mount to view
Some of the other stains used for staining sacchrides include:
- Lectins
- Ruthenium red
- Alcian blue
Immunochemistry
Whereas histochemistry includes a number of techniques used for the visualization of various chemical components in tissues, immunochemistry involves the study of identities and functions of components of the immune system (particularly antibodies).
Essentially, immunochemical methods are based on the selective, reversible and non-covalent binding of antigens by antibodies. This allows for these methods to be used to quantify antigens or antibodies.
All the immunochemical methods/techniques depend on a highly specific and sensitive reaction between antigens and antibodies. There are a number of immunochemistry techniques based on the type of reaction, reagents and samples that are used.
These include:
Particle methods - This is the technique where the antigen-antibody interaction is observed. It includes a number of methods such as Immunoprecipitation, Immunoelectrophoresis, Immunofixation.
Label methods - With label methods, either the antigen or the antibody is labeled allowing for the antigen-antibody reaction to be observed. Immunoassay and competitive binding are examples of label methods.
Some of the other methods include:
- Immunofluorescence
- Immunoelectron Microscopy
Relevant: How does a Microtome work?
Return to Immunofluorescence - In Microscopy, Applications, Direct and Indirect Methods
Return to Immunohistochemistry - Microscopy, Techniques, Cancer Research
Return from Histochemistry to Microscope Research and Information
References
Boguslaw Samotus, Maria Leja, Andrzej Scigalski, Jerzy Dulinski, Robert Siwanowicz (1982) Determination of tyrosine by a modified Millon's reaction and its application to potato tuber extracts.
Culling C.F.A., (1974) Handbook of histopathological and histochemical techniques Ed. 3
Butterworth, London, UK.
Dandekar (1 January 2004). Practicals And Viva In Medical Biochemistry. Elsevier India. p. 28. ISBN 978-81-8147-025-6.
Kiernan. J.A., (1999) Histological and histochemical methods: Theory and practice, Ed. 3
Butterworth Heinemann, Oxford, UK.
Luna, Lee G (1960) Manual of Histologic Staining Methods of the Armed Forces Institute of Pathology (Third Edition). American Registry of Pathology.
Lee Bergma, Stephanie Bechtel and Stefan Wiemann (2006) Immunochemical Methods, Localization.
Symonds DA (1990) Use of the von Kossa stain in identifying occult calcifications in breast biopsies.
Suzuki M, Shinohara Y, Fujimoto T (2013) Histochemical detection of lipid droplets in cultured cells.
Find out how to advertise on MicroscopeMaster!
![Prussian blue stain. Pulmonary veno-occlusive disease. By Yale Rosen [CC BY-SA 2.0 (http://creativecommons.org/licenses/by-sa/2.0)], via Wikimedia Commons Prussian blue stain. Pulmonary veno-occlusive disease. By Yale Rosen [CC BY-SA 2.0 (http://creativecommons.org/licenses/by-sa/2.0)], via Wikimedia Commons](https://www.microscopemaster.com/images/Intra-alveolar_hemosiderin_depositionPrussian_blue_stain.jpg)
![Small cell lung carcinoma with Feulgen Stain By Yale Rosen from USA [CC BY-SA 2.0 (http://creativecommons.org/licenses/by-sa/2.0)], via Wikimedia Commons Small cell lung carcinoma with Feulgen Stain By Yale Rosen from USA [CC BY-SA 2.0 (http://creativecommons.org/licenses/by-sa/2.0)], via Wikimedia Commons](https://www.microscopemaster.com/images/Small_cell_lung_carcinoma-Azzopardi_effect_Feulgen_stain.jpg)