What does a Melanocyte do?
Definition, Function, Life Cycle and Location
Definition
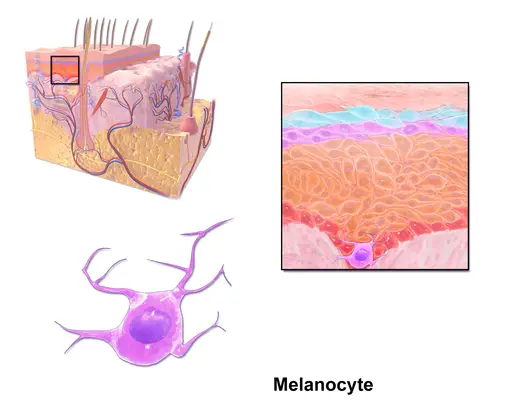
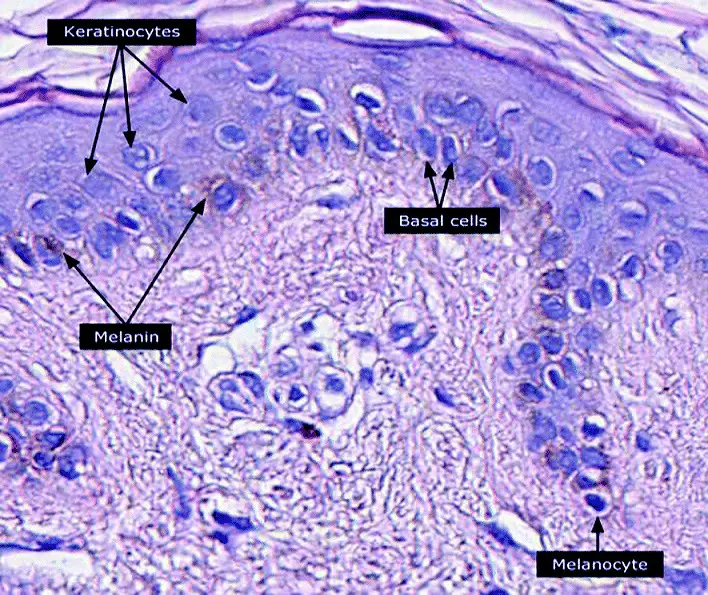
Melanocytes are a group of heavily pigmented cells commonly found in the epidermal basal cell layer. However, they can also be found in other parts of the body including the nervous system and the inner ear, etc.
Although they are primarily involved in photoprotection, through the production of melanin, these cells have also been associated with a number of other physiological functions in different parts of the body.
* Melanocytes are estimated to make up about 8 percent of epidermal cells.
* Because of their ability to produce melanin, melanocytes are the primary components of the pigmentary system- It's worth noting that not all melanocytes produce melanin.
* The number of melanocytes is the same in all races. It's the differences in the amount of melanin produced, size and aggregation of melanosomes that results in the different skin colors between people of different races.
Life Cycle
Melanocytes (pigmented melanocytes) originate from a population of cells known as neural crest cells. These are multipotent cells, capable of self-renewal and differentiation, that originate from the ectoderm germ layer in a developing embryo.
Early on in embryonic development (gastrulation), the neural crest cells (NCC) are produced at the border between neural and non-neural ectoderm known as the neural plate. This occurs under the influence of signalling molecules like Bone Morphogenetic Protein.
Once they are produced, these cells (NCC) go through a process known as epithelial to mesenchymal transition (EMT) which results in the down-regulation of cell adhesion molecules (e.g. E-cadherin). This allows the cells to start migrating. Some of the genes involved in this process include members of the snails/slug family.
This migration is important in that it allows the neural crest cells to differentiate into different types of cells depending on where they localize. For instance, those that migrate and localize at the cranial-caudal axis can differentiate to produce neurons, glial cells, and chondrocytes, etc. while those that localize at the trunk region give rise to endocrinal cells among others. Typically, melanocytes develop from crest cells that localize in the cranial and trunk region (cranial and trunk neural crest cells).
* Based on studies involving mice, the Wnt signaling system was shown to influence the differentiation of NCC to melanocytes.
Whereas cranial neural crest cells give rise to melanocytes that reside in the skin of the head, trunk neural crest cells produce melanocytes found in other parts of the body. However, the trunk neural crest cells are also divided into two populations namely, dorsally and ventrally migrating cells.
Based on a number of studies, some of the melanocytes first originate from the ventrally migrating cells of the trunk NCC. Some transform into neurons while others are retained as multipotent cells. During embryonic cutaneous innervation, these cells migrate to the epidermis.
At the same time, cutaneous nerves undergo branching to produce candelabra pattern. This, along with a number of additional factors, influence the migration and differentiation of melanoblasts that eventually give rise to melanocytes.
Fully differentiated melanocytes reside in the skin, hair follicles, iris, inner ear, choroid of the eye, meninges, and oral mucosa, etc.
* Whereas ventrally migrating trunk NCC (moving between the neural tube and somites) give rise to some cells of the nervous system (e.g. ganglionic cells and Schwann cells) dorsally migrating trunk NCC (moving between somites and surface ectoderm) only give rise to melanocytes that localize in the epidermis and hair.
* Epidermal melanocytes remain in communication with the nervous system.
Melanoblast/Melanocyte Characteristics
The melanoblast is the precursor cell that gives rise to melanocytes.The melanoblast itself originates from the neural crest cells. Unlike melanocytes, a melanoblast is incapable of producing melanin. This is due to the absence of the enzyme Tyrosinase which catalyzes the process through which this pigment is made.
As part of the neural crest cells, melanoblasts have to migrate in order to reach specific destinations. This movement has been shown to involve an interaction with the extracellular matrix and keratinocytes. Here, movement usually involves the extension of dynamic pseudopodia as the cell squeezes through the epidermal keratinocytes using myosin motors.
* With respect to morphology, melanoblasts are elongated cells with long protrusions that traverse the spaces between neighboring keratinocytes.
Following the differentiation of melanoblasts into melanocytes, they undergo maturation with the ability to produce melanin. Mature melanocytes are characterized by a number of specific proteins including Tyrosinase, tyrosinase-related protein 1 and 2, and melanosomal matrix proteins, etc. Morphologically, they are smaller than keratinocytes and have an oval shape.
* Melanocytes are also fewer in number when compared to keratinocytes (1:10).
* There are an estimated 1,200 melatinocytes per mm2 of the skin.
Melanocytes are also characterized by organelles known as melanosomes where melanin is produced. Like keratinocytes, they also possess dendritic processes that transfer melanin to the former.
* With respect to hair follicles, melanocytes are located at the proximal bulb where they are referred to as bulb melanocyte. In addition, they can also be found in the sebaceous gland.
Melanocytes
Skin Anatomy
Before looking at melanogenesis, the process through which mature melanocytes produce melanin, it's important to understand the skin anatomy.
Depending on the region of the body, the skin measures between <0.6mm (skin of the eyelids) and 2.0mm (e.g. soles of the feet) in thickness and makes up about 15% of the total body weight in adults.
Generally, the skin consists of three main layers that include:
Epidermis - The epidermis is the outermost layer of the skin. Also known as stratified squamous epithelium, this layer consists of several layers of keratinocytes.
These include:
Stratum basale - Also known as the basal layer, stratum basale is the innermost layer of the epidermis. As such, it consists of continually dividing cells that replace cells of the layers above. Some of the other cells located in this region include melanocytes and Market cells.
Stratum spinosum (spiny layer) - This is the thickest layer and consists of squamous cells (prickle cells) and is located above stratum basale. Here, cells produce keratin, a tough protein that can also be found in nails and hair. Langerhans cells (antigen-presenting cells) can also be found in this layer.
Stratum granulosum - Also known as the granular layer, the Stratum granulosum consists of flattened keratinocytes. A good number of cells in this layer do not have nuclei.
Stratum lucidum - Cells in this layer are also flattened and dehydrated. They are also more fused together. These cells do not have nuclei.
Stratum corneum (horny layer) - The stratum corneum consists of mostly dead cells. This being the outermost layer, cells are continually lost and replaced by cells of the layer below it. These cells also lack a nucleus.
Dermis
The layer beneath the epidermis is known as the dermis. It's separated from the epidermis by the basement membrane.
It's the thickest part of the skin and consists of several important structures that include:
Blood vessels - Supply oxygen and nutrients and remove waste products
Lymph vessels - supply lymph which contains immune cells
Hair follicle - Sheath that surrounds the base of the hair - It serves to nourish the hair
Sweat glands - Secrete sweat which not only regulates body temperature but also promotes bacterial growth
Sebaceous gland - Attached to the hair follicle and produce oil (sebum) to keep the skin smooth and protect the skin
Nerve endings - Receptors that transmit sensations related to touch, pain, itch, and pressure, etc.
Hypodermis/Subcutis - This is the third and innermost layer of the skin. It is largely composed of fat and collagen cells.
It serves a number of important functions including:
- Insulation
- Absorbing shock
- Protecting inner organs
Melanogenesis
As mentioned, melanogenesis is the process through which mature melanocytes produce melanin. Compared to cells in the upper layers of the epidermis, cells in the basal layer have nuclei. Therefore, exposure to UV light can cause DNA damage in these cells.
In the event of DNA damage due to these rays (E.g. exposure to sunlight at the beach without using sun cream), DNA damage response results in a cascade of events that end with increased production of melanin.
The following are the step/events that lead to increased production of melanin in the skin following DNA damage:
· p53 stimulation - This is a transcription factor that is produced in the event of DNA damage
· POMC expression - p53 influences increased expression of the protein POMC (Pro-opiomelanocortin). Pro-opiomelanocortin is the precursor to several hormones including Alpha-MSH (Alpha melanocyte-stimulating hormone)
· Alpha-MSH is then released from the keratinocyte and binds to the receptor MC1R (melanocortin 1 receptor) of the melanocyte cells. Here, it stimulates the expression of enzymes and proteins required for melanin within the melanosomes
· Extended arms/pseudopodial extensions of the melanocytes (which are in contact with keratinocytes) allow melanosomes to move into the keratinocytes
· In the keratinocytes, the melanosomes degranulate to release their contents (melanin) which in turn surround/shield the nucleus
· As they surround and shield the nucleus, the melanin absorbs the UV light rays and convert this energy to heat through a process known as internal conversion. This protects the DNA from being damaged by UV rays which can cause mutations
* While the number of melanocytes is the same for people of all races, the amount of melanin produced varies depending on exposure to sunlight. For instance, because people from Africa and parts of the middle east are exposed to more sunlight than those in European countries, their skin is darker due to high melanin production.
* Melanin is commonly found in keratinocytes of the stratum spinosum and stratum basale. Compared to the cells in the upper layers, these cells have nuclei and need melanin for protection.
Proliferation, Life Span, and Death
Compared to melanoblasts, melanocytes are highly differentiated and specialized. However, based on a number of studies, only a few of these cells are capable of division. Here, differentiation and specialization of the cells has been shown to reduce their proliferative potential.
While epidermal melanocytes are long-living cells, between 10 and 20% are lost every decade after an individual reaches 30 years of age. For this reason, the total number of melanocytes starts decreasing after 30 years of age. In addition to this decrease, they have also been shown to start changing their shape during his period. For the most part, this involves an increase in size and more dendritic processes.
* Unlike epidermal melanocytes, the life span of hair melanocytes is largely dependent on the life cycle of hair: 3 to 5 years.
Generally, the decrease in the total number of melanocytes is a result of apoptosis (programmed cell death). Here, the process has been shown to be influenced by the gradual accumulation of reactive oxygen substrates. These substances accumulate due to decreased activity of the enzyme catalase.
While some of the cells are capable of division/proliferation, this is highly limited to terminally differentiated melanocytes. This is because the nitrogen-activated protein kinase pathway involved in this process is not active in these cells.
Retinal Pigment Epithelial (RPE)
The retinal pigment epithelial is a monolayer of cuboidal cells located between the photoreceptor cells and choroid (a thin, vascular middle layer of tissue in the wall of the eye). While the retinal pigment epithelial cells have a different origin from melanocytes, they are also involved in the production of melanin.
In addition to a cuboidal morphology, they are also characterized by apical microvilli and basal infoldings which enlarge the surface.
As well, melanosomes, which are oval in shape, are usually located near the microvilli. One of the main functions of retinal pigment epithelial is to protect cells from damage. Here, studies have shown the melanin produced helps to protect the cells from oxidative stress damage. The melanin pigment achieves this by acting like a free radical sink thus reducing the overall cytotoxic lipid peroxidation.
* The retinal pigment epithelial also acts like a selective barrier to the photoreceptor layer and also as a vegetative regulator.
* The choroid also produces melanin.
Functions
Melanocytes are involved in several functions that include:
Cutaneous pigmentation - Cutaneous pigmentation is the result of two main events; production of melanin granules and their transfer to keratinocytes in the layers above. The color of the skin is largely dependent on the amount of melanin produced.
Protection from UV rays (ultraviolet radiations) - As already mentioned, the melanin produced in melanocytes surround the nucleus of neighbouring keratinocytes thus protecting the nucleus from damage - When melanocytes start growing out of control, this can result in melanoma.
Eye color - The quantity of intracellular melanin produced by melanocytes in the uveal tract of the eye determines the eye color. In addition to the quantity of melanin, some of the other factors that influence the color include the type of melanin and how it is packaged within the melanosomes in this region.
Some of the other functions associated with melanocytes include:
· They are intermediate cells in the stria vascularis of the cochlear. Here, they have been shown to initiate endolymph-mediated action potentials required for normal hearing.
· In the inner ear, melanin granules are also suggested to promote body balance.
· In the brain, a type of melanin known as neuromelanin is suspected of playing an important role in defense by binding and eliminating reactive oxygen species.
Return from What does a Melanocyte do? to MicroscopeMaster home
References
J.R. Sparrow, D. Hicks, and C.P. Hamel. (2014). The Retinal Pigment Epithelium in Health and Disease.
Mirosława Cichorek, Małgorzata Wachulska, Aneta Stasiewicz, and Agata Tymińska. (2013). Skin melanocytes: biology and development.
P. M. Plonka et al. (2009). What are melanocytes really doing all day long…?
Sharique A. Ali and Ishrat Naaz. (2015). Current Challenges in Understanding the Story of Skin Pigmentation — Bridging the Morpho-Anatomical and Functional Aspects of Mammalian Melanocytes.
Links
https://www.sciencedirect.com/topics/medicine-and-dentistry/melanocyte
Find out how to advertise on MicroscopeMaster!