What is the Function and Location of Glial Cells?
Vs Neurons
First described in 1838 by Robert Remak, an embryologist and neurologist, Glial cells (also known as Glia or neuroglia) are cells of the nervous system (central and peripheral nervous system) other than neuronal cells.
Although they are not involved in information processing which is the central function of neurons, glial cells make up the majority of the cells of the nervous system and provide the support required by neurons to effectively perform their functions. Therefore, neurons would not be able to function in the absence of glial cells.
In both the central and peripheral nervous systems, there are several types of glial cells which include:
- Astrocytes
- Ependymal cells
- Oligodendrocytes
- Microglia
- Satellite cells
- Schwann cells
* The word Glial is derived from the Greek word "glia" meaning glue.
Where are Glial Cells found?
While glial cells are different from neurons and also perform several functions, they are closely related in that they originate from the same population of stem cells.
In the central nervous system, studies have shown that glial cells originate from stem cells located in the ventricular zone in the neural tube while those in the peripheral nervous system are produced by progenitor cells located in the neural crest.
Following their development, these cells vary in comparison to neurons not only in their functions, but also in shape, size, and physiology. Considering the diversity of glial cells, they can also be found in different parts of the body.
Glial Cells in the Central Nervous System
The central nervous system consists of the brain and the spinal cord. There are three types of glial cells that can be found in the central nervous system.
These include:
Astrocytes - in the central nervous system, astrocytes are found near the neurons where their radiating processes wrap around the neurons
Oligodendrocytes - shown to line up along the nerve fibers in the central nervous system
Microglial cells (microglia) - found near neurons and may even be in touch with neighboring neurons
* A fourth type of glial cells in the central nervous system is known as ependymal glia. These cells line the ventricles of the central nervous system in the brain parenchyma.
Glial Cells in the Peripheral Nervous System
The peripheral nervous system is part of the nervous system that consists of nerve cells and ganglia outside the central nervous system. As such, it serves to connect various parts of the body (the skin, body organs, and limbs) to the central nervous system. There are two main types of glial cells located in the peripheral nervous system.
These include:
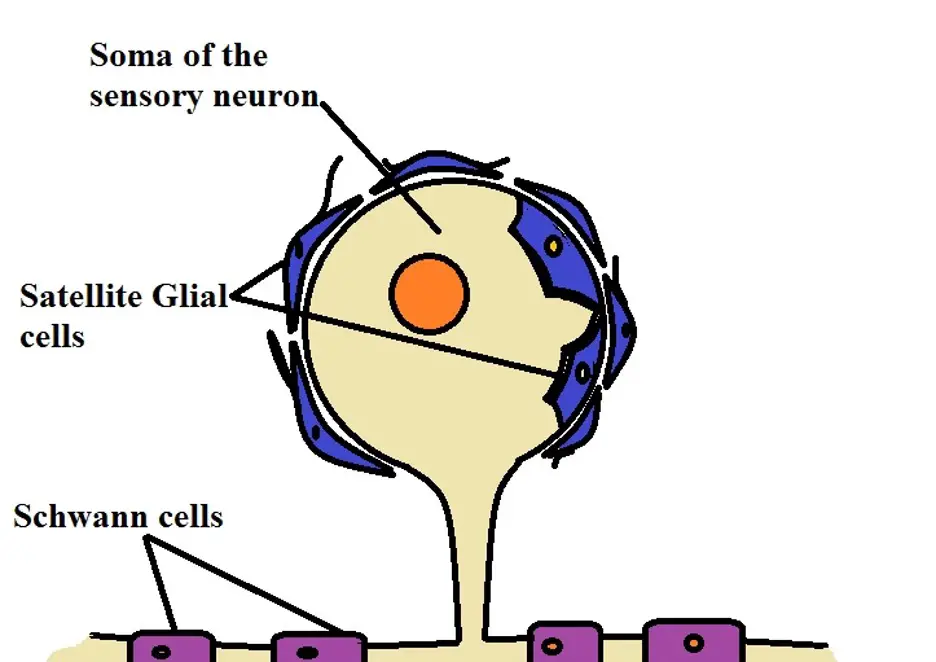
Satellite cells - In the peripheral nervous system, satellite cells (satellite glia) are located in the ganglia where they are positioned near the neuronal somata.
Schwann cells - Unlike satellite glia, Schwann cells are typically found in close proximity with axons.
Types and Functions
As mentioned, there are different types of glial cells in the central and peripheral nervous system. While they are collectively known as glial cells, they have different functions which contribute to the primary functions of the neurons.
This section will focus on the different types of glial cells and their respective functions:
Astrocytes
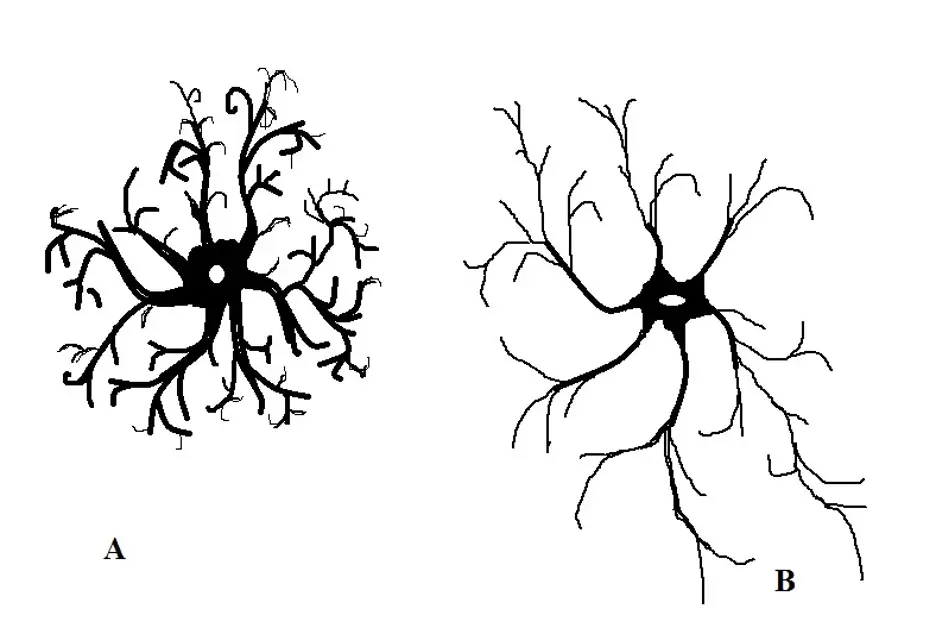
Characterized by a stellar morphology (star-shaped), astrocytes are the most abundant glial cells in the central nervous system and particularly in the brain. While they are the most abundant cells in the brain, astrocytes can be found in all parts of the central nervous system.
Here, however, it's worth noting that there are two main types of astrocytes, based on morphology, that include:
Protoplasmic astrocytes - Located in the gray matter surrounding the synapses, protoplasmic astrocytes are generally shorter in size with pudgy processes. Based on staining techniques, protoplasmic astrocytes have been shown to be morphologically complex with many fine processes (branching).
In the gray matter, where these cells are uniformly distributed, protoplasmic astrocytes also extend to neighboring blood vessels and wrap around them which plays an important role in their functions.
Some of the other characteristics of protoplasmic astrocytes include:
- Possess fewer fibrils
- Cytoplasmic organelles within the cells are sparsely distributed
- The processes/branches of protoplasmic astrocytes are shorter compared to those of fibrous astrocytes
Fibrous astrocytes - Unlike protoplasmic astrocytes, fibrous astrocytes are distributed in the white matter. Generally, these cells are less complex (compared to protoplasmic astrocytes) with less branching. Although they have less branching processes, they are relatively longer.
In the white matter, fibrous astrocytes are longitudinally oriented along with the fiber bundles. Like protoplasmic astrocytes, fibrous astrocytes also have fewer and sparsely distributed organelles. In addition, some of their processes consist of vascular feet that wrap around neighboring blood vessels (capillaries).
a- Protoplasmic astrocytes
b- Fibrous astrocytes
Functions
In addition to being some of the most abundant glial cells in the central nervous system, astrocytes are also involved in several important functions that include:
Blood-brain barrier - As mentioned, the highly branched protoplasmic astrocytes have vascular feet that encircle the neighboring blood vessels particularly the capillaries. In doing so, they form the glial limiting membrane which is the basis for the outermost wall of the blood-brain barriers.
This is important for the brain in that it prevents large substances in the blood from entering the extracellular environment of the brain.
While the barrier creates a separation that keeps the brain in a separate compartment, it ensures that blood vessels deliver materials required by cells of the central nervous system (oxygen, etc). At the same time, it keeps unwanted substances away.
Support - With their cytoplasmic processes, astrocytes also provide structural support for the neurons and are therefore directly involved in the general structure of the brain. While proving support for differentiating neurons, they are also involved in the myelination process and thus to the organization of these cells.
Homeostasis - In the central nervous system, both protoplasmic and fibrous astrocytes are involved in homeostasis. Here, studies have shown the two types of astrocytes to help in the regulation of ionic concentration by maintaining a healthy ionic environment. In doing so, they regulate the generation as well as transmission of impulses in the neurons.
* At the synapses, protoplasmic astrocytes extend their processes which are involved in the uptake of neurotransmitters at the synaptic cleft thus contributing to the transmission of information.
Waste removal - In the central nervous system, astrocytes have been shown to collect various metabolites among other waste material from the cells and the extracellular matrix and deliver them to the surrounding capillaries so that they can be removed from this environment.
This is an important function of astrocytes which ensures that harmful materials are excreted thus enhancing the functions of neurons.
Energy reserve - Astrocytes form a bridge between the vascular system and neuronal synapses. This is an important anatomical setup in that astrocytes have been shown to increase the delivery of blood to regions where neurons are active.
By increasing blood supply to these regions, astrocytes promote the delivery of materials (e.g. oxygen, etc) that ensure that neurons continue functioning properly.
As well, astrocytes act as energy reserves because they contain glycogen that is broken down during hypoglycemia. By storing this energy source, astrocytes can release it so that it can be broken down to provide the energy required by neurons when need be.
In addition to the functions above, new research has shown that astrocytes also have the following functions:
- Formation of long term memory
- Repair of neuronal tissues through the formation of a glial scar
- Formation of cellular network through the communication of astrocytes via gap junctions
Oligodendrocytes
Oligodendrocytes, which are also located in the central nervous system, were first described as a distinct type of glial cells in 1919.
As compared to some of the other glial cells, oligodendrocytes have fewer processes which is one of their defining characteristics. Like the other glial cells, they have sparsely distributed organelles. Because of the small size of the cell body, the spherical nucleus occupies a large volume of the cell.
In addition, they also contain an abundant amount of endoplasmic reticulum involved in myelin synthesis. While oligodendrocytes contain fewer processes compared to some of the other glial cells, the few processes that are there encircle/wrap around the axons of the neurons in the central nervous system.
Functions
While oligodendrocytes are commonly referred to as myelinating oligodendrocytes, some of these cells are non-myelinating.
For this reason, oligodendrocytes are divided into two main types that include:
Myelinating Oligodendrocytes - This type of oligodendrocytes is commonly found in the white matter of the central nervous system. As the name suggests, these cells are involved in the myelination of nerve fibers/neurons.
Using protein and fatty substances, oligodendrocytes are able to synthesize the myelin sheath of neurons which promote the rapid transmission of electrical impulses along the nerve cells.
There are several types of myelinating oligodendrocytes which include:
· Type I myelinating Oligodendrocytes - Form myelin segments characterized by diverse orientation/arrangement
· Type II myelinating Oligodendrocytes - Myelin segments formed by type II cells are arranged parallel to each other
· Type III myelinating Oligodendrocytes - Unlike Type I and Type II cells, these cells generally form a small number of segments on axons with a large diameter
* Myelination of axons of neurons in the white matter is a highly regulated process dependent on the differentiation process (of both the neurons and oligodendrocytes) as well as signaling from the central nervous system.
* Myelinating oligodendrocytes are also involved in the repair of myelin in the event of damage.
Non-myelinating Oligodendrocytes - Unlike myelinating oligodendrocytes, non-myelinating oligodendrocytes are commonly found in the grey matter of the central nervous system.
As the name suggests, these cells are not involved in the synthesis of sheath/myelin around the axons of neurons. Rather, they play an important role in controlling the extracellular matrix around nerve cells in the grey matter.
* The disruption of myelin in the central nervous system has been associated with diseases like multiple sclerosis and the degeneration of axons etc. For this reason, oligodendrocytes are essential for the normal functioning of neurons.
Microglial cells
Like oligodendrocytes, microglial cells (microglia) were also identified as distinct glial cells in 1919. Like the other two cells above microglial are located in the brain and spinal cord (central nervous system).
While they have a small cell body, like oligodendrocytes, microglial cells also consist of fine, branched processes on their surface. Like many other cells, microglia also consists of a spherical, central nucleus and a number of other cell organelles.
While microglial cells are generally described as having a relatively small cell body and branching processes, the general morphology changes depending on the state of the cells.
In the resting state, microglia tend to be highly ramified which means that they are highly branched. However, the cell body becomes larger in size when they are in an activated state as the surface processes contract. This activated stage is also characterized by increased proliferation as they prepare to perform their function.
In the phagocytic stage, on the other hand, they are characterized by an amoeboid morphology and are capable of producing several immuno-molecules.
Functions
In the central nervous system, as already mentioned, microglial cells act as the tissue-resident macrophages; As such, they are the main innate immune effector cells in this region of the nervous system.
Following activation, in the event of pathological conditions, they transform to macrophages involved in the removal of cellular debris as well as any dead neurons through phagocytosis.
In addition to their primary function in establishing a protective environment in the central nervous system, recent studies have shown that microglial cells also have physiological functions.
Under normal conditions, these functions involve synapse monitoring, phagocytosis of synapses during their development, as well as phagocytosis of adult-born neurons (regulated activities). Here, the disruption of these activities has been shown to result in developmental disorders.
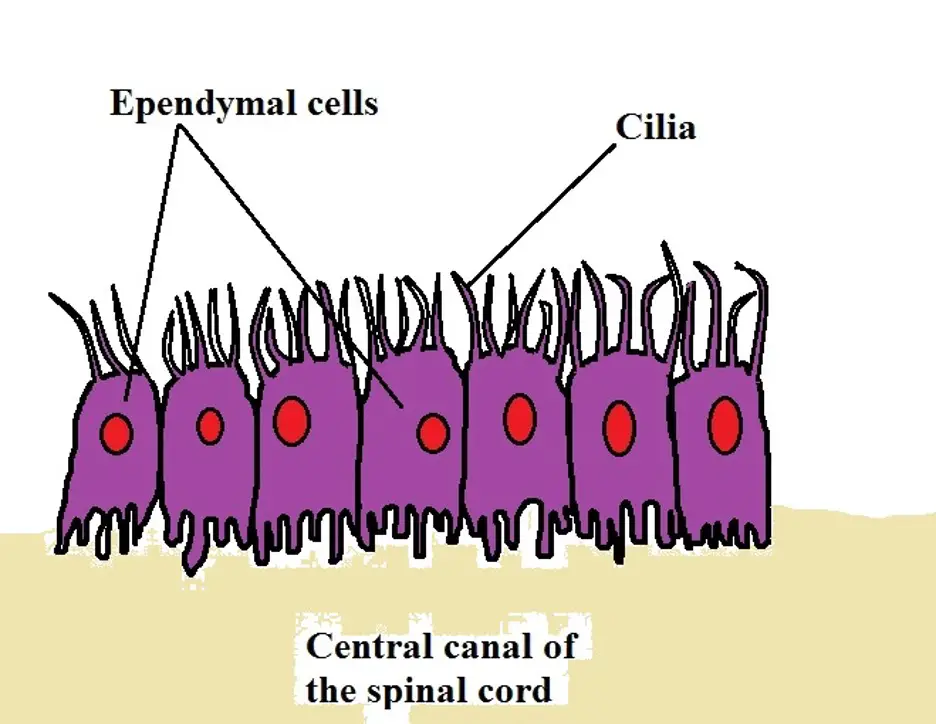
Ependymal glial cells
Like the other glial cells highlighted above, ependymal cells are also located in the central nervous system. Here, they can be found lining the ventricles of the brain as well as the central canal of the spinal cord. Apart from these regions, they can also be found in a network of blood vessels known as the choroid plexus.
In tissue culture, ependymal cells have been shown to exhibit morphological diversity, ranging from round and columnar to triangular and cone or spindle-shaped. However, in vivo, they are generally cuboidal or columnar in shape.
They are also different from the other cells of the central nervous system in that they possess cilia and microvilli on their surface.
* In fetus, ependymal glial cells are characterized by long basal processes.
Functions
Depending on the stage of development, ependymal glial cells have a number of functions that include:
· In the brain of a mature individual, they are involved in the transport of electrolytes as well as solutes between the cerebrospinal fluid and brain parenchyma
· In addition to circulating cerebrospinal fluid, they also absorb it
· In the fetus, they are involved in the secretion of various molecules including glycosaminoglycans which prohibit the growth of axonal cones
· They also stop mitosis in the germinal matrix in the fetus
Satellite glial cells
Satellite glial cells are a type of glial cell found in the peripheral nervous system. In particular, they have been identified in the sensory, parasympathetic, and sympathetic ganglia in the peripheral ganglia.
With regards to morphology, satellite glial cells are thin and flattened given that they cover/surround the soma sensory neurons. In general, then, they are arranged in a manner that resembles the outer covering of a soccer ball.
* For the most part, satellite glial cells form a complete envelope around the somata.
Functions
As mentioned, satellite glial cells can be found covering the surface of neuron cell bodies. As such, they are among the glial cells that are in the closest proximity with neuron cell bodies.
Here, they have a number of important functions that include:
- Supplying nutrients to the neurons and regulate neuronal homeostasis
- They influence the structure and structural functions of neurons
- By covering the cell bodies of neurons, they also have a protective function
Difference between Neurons and Glial Cells
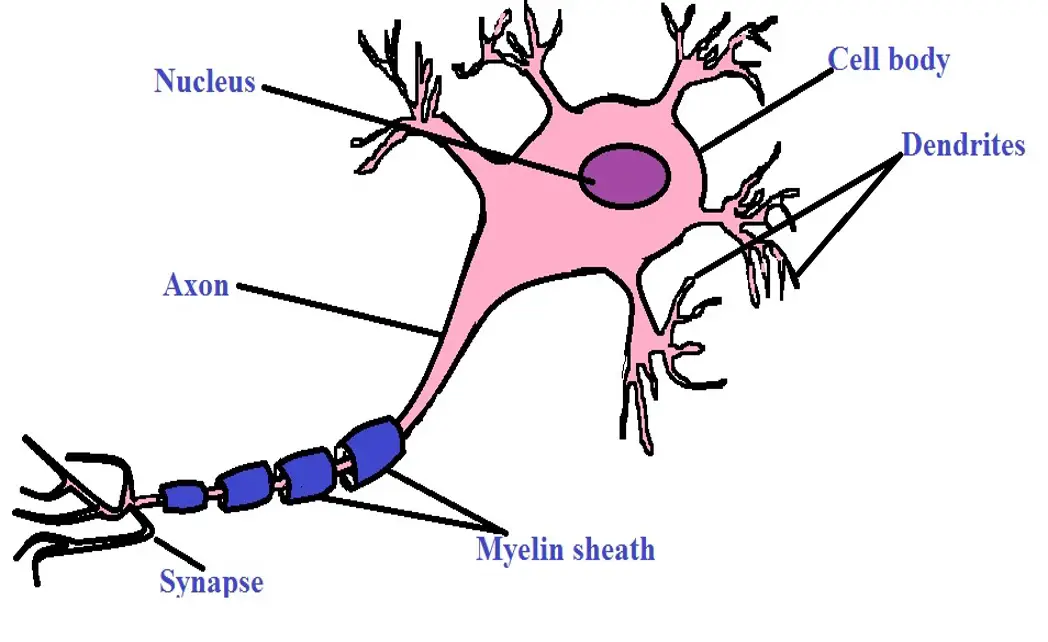
Neurons are specialized cells of the nervous system that send and receive information from the brain. It's through this activity that an individual (animal, human being, and other organisms) is able to respond appropriately to different stimuli.
Glial cells, on the other hand, are important cells of the nervous system that promote the functions of neurons by providing structural support, nourishment, and protection, etc. While they have several differences, they are both cells of the nervous system.
The following are some of the main differences between neurons and glial cells:
Functions
As mentioned, neurons are primarily involved in the transmission of nerve impulses and thus the transmission of information to and from the brain. This is because they are excitable and can transmit electric impulses.
Glial cells, on the other hand, are incapable of transmitting nerve impulses and are specialized to support neurons and their functions.
Number/population - While there are many neurons (between 86 and 100 billion) in the human brain, the number of glial cells is much larger, ranging from 5 to 10 times the number of neurons - This is due to the fact that there are different types of glial cells with different functions.
Morphology - While glial cells have processes that extend from the cell bodies, they are structurally different from neurons in that they lack axons and dendrites which are characteristic of neurons. Moreover, they are generally smaller in size when compared to neurons.
Although neurons also vary in general morphology, they are all characterized by a cell body, dendrites, and axons. Some of the neurons are myelinated while others are unmyelinated.
See also: Stem Cells, Somatic Cells, Neural Progenitor Cells
Return to Cell Biology main page
Return to learning about Nerve Cells
Return from "What is the Function and Location of Glial Cells?" to MicroscopeMaster home
References
Alexei Verkhratsky and Arthur Butt. (2013). Neuroglia: Definition, Classification, Evolution, Numbers, Development: Glial Physiology and Pathophysiology, First Edition.
Beckstead R.M. (1996) Glial Cells and Their Functions. In: A Survey of Medical Neuroscience.
Hiroaki Wake and R. Douglas Fields. (2015). Physiological Function of Microglia.
Sarah Jäkel and Leda Dimou. (2017). Glial Cells and Their Function in the Adult Brain: A Journey through the History of Their Ablation.
Siegel GJ, Agranoff BW, and Albers RW, et al. (1999). Basic Neurochemistry: Molecular, Cellular and Medical Aspects. 6th edition.
Links
Find out how to advertise on MicroscopeMaster!