Chlorophyll
** Definition, Structure, Function, and Photosynthesis
Definition: What is Chlorophyll?
Chlorophyll is perhaps the most important naturally occurring pigment on the planet. Found in plants and some microorganisms (e.g. cyanobacteria), these porphyrins play an important role in the conversion of solar energy to chemical energy through a process known as photosynthesis.
The different forms of chlorophyll are essential for life given that they are responsible for the autotrophic nature of plants and some microorganisms.
Apart from photosynthesis, chlorophylls are also used for a number of commercial purposes in agriculture and the food industry.
* Plants and microorganisms that contain chlorophyll may be described as photosynthetic organisms.
See also: Photosynthetic Bacteria
* The term chlorophyll was coined in 1818 by French Pharmacists; Pierre Joseph Pelletier and Joseph Bienaime Caventou.
* The word chlorophyll comes from two Greek words; Chloros which means green and phyllon which means leaf.
The different forms of chlorophyll include:
- Chlorophyll a
- Chlorophyll b
- Chlorophyll c1
- Chlorophyll c2
- Chlorophyll d
- Chlorophyll f
Structure of Chlorophyll
Like heme groups, chlorophylls are porphyrins found in plants. As such, they tetrapyrroles that contain a metal ion at their core. Unlike heme groups, which contain iron at their core, the metal ion found in chlorophyll is magnesium.
Here, biosynthesis of the pigment starts with the two important intermediates (uroporphyrinogen III and protoporphyrin IX) followed by the insertion of magnesium ion at its core. However, further modifications result in a variation of the different forms and specialization of the pigment in different organisms.
Chemically, then, chlorophyll is composed of the following components: A nucleus of porphyrin (tetrapyrrole) that contains a chelated magnesium atom - The porphyrin head of the structure is composed of four rings of pyrrole with nitrogen arranged in a ring
Hydrocarbon chains linked through a group of carboxylic acid - The long hydrocarbon chain makes up the tail part of the structure.
* Chlorophyll pigments are bound by proteins that hold them in the right position. This positions them in the right alignment to trap light energy and transfer it during photosynthesis.
* Chelate - A molecule (organic molecule) that contains a metal ion at its core.
* Whereas chlorophyll a contains a methyl constituent in its structure, this is replaced by a formyl group in chlorophyll b.
* In nature, tetrapyrroles possess a structure that is essential for important biological processes.
* Being some of the most common tetrapyrroles in nature, heme groups and chlorophylls are commonly referred to as the "Colors of life".
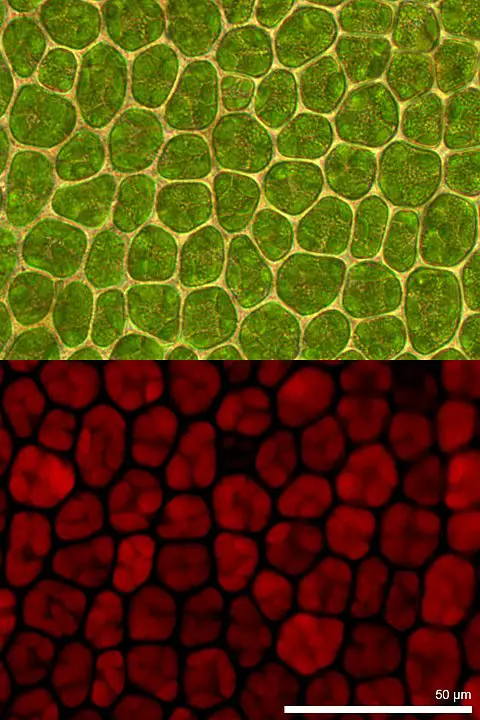
In plants, the pigment is located in Photosystems I and II within the thylakoid membrane of chloroplasts. This, then, means that chlorophylls are membrane-bound pigments in plants (consisting of eukaryotic cells).
Here, chlorophyll pigments perform their functions within the chloroplast. Although they are membrane-bound, in that they are confined in the chloroplast, this also helps enhance their functions.
For some such prokaryotes as cyanobacteria capable of photosynthesis, however, the chloroplast (as well as other membrane-bound organelles) is absent. For this reason, the pigments are not enclosed in a plasma membrane as is the case with eukaryotic cells.
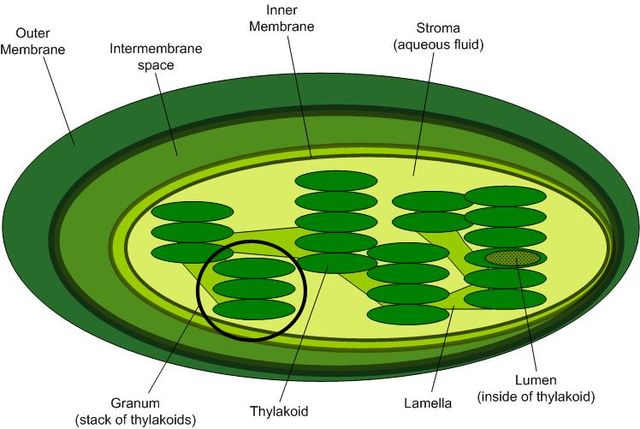
Brief Description of the Chloroplast
In plants, chloroplasts are organelles found in cells of the mesophyll layer; where photosynthesis takes place. Like some of the other organelles (e.g. nucleus and mitochondria) of eukaryotic cells, chloroplasts are surrounded by a double membrane that separates the inner parts of the organelle from the intracellular environment.
Within the chloroplast, another membrane forms disc-shaped structures known as thylakoids (which forms the thylakoid system) within which the pigments (chlorophyll) are located.
Some characteristics of chloroplasts include:
- Have a double membrane that is permeable to small molecules and ions
- Are biconvex or planoconvext in shape
- Range between 4 and 6 µ in diameter
- In some organisms (particularly algae) the organelles have been shown to form a network
Chloroplast Structure
Green Chlorophyll
Chlorophyll gives plants (and some organisms) their green color.
To understand why plants appear green to the human eye, it's worth noting that unlike some of the other animal groups, human beings are trichromats and thus interpret colors depending on the ratio of three primary wavelengths of the visible light (red, green and blue light).
In nature, chlorophyll, which is green in color, is capable of absorbing the red and blue lights within the visible spectrum of light. This, however, is not the case with green light which is reflected away. As a result, the brain interprets the color as green given that this is the color that is reflected by chlorophyll.
Unlike human beings, some animals are capable of distinguishing between more basic wavelengths of light (e.g. birds which are tetrachromatic) and can, therefore, differentiate between more colors.
Although it's not possible to actually tell how objects in the physical world appear to some animals (e.g. birds and insects) studies have shown that color vision is more effective for some of these organisms.
For instance, whereas birds have been shown to be capable of distinguishing more hues of green as compared to human beings. some insects are capable of seeing ultraviolet light.
* As plants prepare for winter (in fall) the production of chlorophyll ceases. Any chlorophyll available also breaks down which in turn causes the green color to fade away.
As a result of this decomposition and the presence of other pigments (e.g. anthocyanin in some plants) plants may start appearing more yellow/orange/reddish etc in color.
* Change in color is also the result of chlorophyll residues mixing with the other pigments.
Forms/Types of Chlorophyll
Chlorophyll A and B
For a majority of organisms capable of photosynthesis (plants, cyanobacteria, algae) chlorophyll a is the primary pigment of photosynthesis. It's responsible for absorbing light in the orange to red and violet to blue spectrum that provides the energy required for subsequent photosynthetic reactions.
While chlorophyll a is essential for photosynthetic organisms, this is not the case with chlorophyll b. Although it is one of the main chlorophyll pigments, not all photosynthetic organisms contain this pigment. As such, it serves as the accessory pigment to chlorophyll a.
As compared to chlorophyll a, chlorophyll b absorbs blue light and thus serves to expand the spectrum of light absorbed. For plants with chlorophyll b, then, a higher range of energy from the sun is converted to chemical energy as compared to plants that lack the pigment.
According to studies, the presence of chlorophyll b is one of the adaptive strategies that allow plants in environments with less sunlight to absorb a wider range of light wavelength.
* While Chlorophyll a and b are similar in structure (in that they have a tadpole-like shape) the structural difference between the two is as a result of a single atom on the third carbon in the side chain at the porphyrin ring (head of the structure).
* Whereas Chlorophyll has a blue/green color, chlorophyll b has an olive green color.
In all plants capable of oxygenic photosynthesis, Chlorophyll a (Chl a) is not only involved in the light-harvesting complexes, but also in reactions that occur in the reaction centers. As a complement pigment, Chlorophyll b (Chl b) serves to harvest light at the ratio of 1:3 (to Chl a).
It can be found in green plants and algae as well as organisms like prochlorophytes. In some organisms (e.g. some chromophytes) the pigment has is accompanied by c-type chlorophylls.
* In plants and microorganisms with chlorophyll b, the pigment is located in antenna complexes of PS II (in the light harvesting complexes) where it makes up about 50 percent of chlorophyll.
Chlorophyll C
Chlorophyll c (Chl c) can be found in a number of organisms including nine Divisions of algae (chromophyte algae) as well as two Divisions of prokaryotes.
Depending on the organism, this chlorophyll co-occurs with other pigments (e.g. Chl a and carotenoids). It's also different in structure compared to some of the other chlorophylls.
For instance, as compared to the other forms of chlorophyll (which are Mg-chlorins), chlorophyll c has been shown to be Mg-phytoporphyrins. At the C-17 ring, the structure of this pigment contains propenoic acid (a transacrylic acid) that replaces the propionic acid side chain found in chlorophylls a and b.
With regards to the structure, forms of chlorophyll c that are polar in nature are protochlorophyllides (precursors of chlorophyll). However, due to their ability to harvest light, they are regarded as chlorophyll.
Variations of chlorophyll c identified so far include:
- Chl c1
- Chl c2
- Chl c3
- Chl c(cs-170)
* In its natural state, Chl c has brownish-golden color.
* For the most part, Chl c pigments are found in a wide range of marine and freshwater algae (as well as some microorganisms).
Chlorophyll d
Found in red algae and some microorganisms (cyanobacteria), chlorophyll d is a minor pigment that is involved in the capture of the red spectrum of light (far end spectrum of red light). With regards to structure, Chl d is also different from some of the other forms of chlorophyll.
For instance, in comparison to chlorophyll a, chlorophyll d has formyl group (Here, the formyl group takes the place of a C3-vinyl group located on the chemical structure of Chl a). For such organisms as A. marina, high percentage of the chlorophyll makes it possible for them to thrive in environments with less sunlight.
According to a study on A. marina, Chl d was shown to help the organism absorb light that had been depleted of the red and blue wavelengths of light. Here, the organism was still able to absorb near-far-red wavelength of light for survival.
Chlorophyll f
Chlorophyll f is the most recent form of chlorophyll to be identified. Unlike the other pigments found in plants and microorganisms, Chl f is found in wet lands cyanobacteria. Like Chl a and c, however, Chl f has also been shown to be an accessory pigment in the antenna system of cyanobacteria.
One of the biggest advantage of the pigment, as compared to the others, is the fact that it is capable of absorbing high amounts of near-infrared light as compared to the other forms of chlorophyll. As such, it gives the organism a competitive edge in that energy is harvested from a different light wavelength as compared to other organisms that rely on light for photosynthesis.
In biological sciences, this was an important discovery given that oxygen producing microorganisms (during photosynthesis) were not thought to be capable of using infrared light as a source of energy.
Role of Chlorophyll in Photosynthesis
Photosynthesis is arguably the most important process on our plant in that it is the basis of life. During photosynthesis, energy from the sun is used to convert carbon dioxide and water to organic molecules that not only allow plants to continue growing and reproducing, but also serve as food sources for various animals and human being.
In general, photosynthesis may be divided into two main phases, namely, light/energy-harvesting phase (where chlorophyll captures light energy) and the carbon reduction phase (the Calvin cycle).
How Chlorophyll Captures Light Energy
Depending on an object, some wavelengths of light are absorbed while others are reflected which allows us to see and differentiate colors. For the most part, most objects heat up as they absorb light energy. This, however, is not the case with chlorophyll.
While green light is reflected, thus giving off the green characteristic of plants (especially leaves), chlorophyll absorbs the red and blue light which in turn excites some electrons in the ring of the pigment. The image below highlights excited electrons when the pigment absorbed light energy.
Here, then, sunlight (photon of light) provides the energy required to energize electrons which in turn breaks free from a chlorophyll atom and enters the electron transfer chain. Given that electrons required for the process are produced by the pigment, chlorophyll, in this case, is referred to as an electron donor.
* Electrons donated by the chlorophyll is replaced when a water molecule is split.
* Energy between neighbouring pigments in a photosystem is transferred through a process known as resonance energy transfer (a form of electromagnetic interactions)
Following excitation, an electron in Photosystem II is passed down the electron transport chain before reaching the final electron acceptor. Here, the electron is transferred to an organic molecule (plastoquinone), a cytochrome complex and ultimately a protein known as plastocyanin (which contains copper).
Electron transport through this chain causes the change from a higher to lower level of energy with some of this energy being used to pump hydrogen protons from the stroma.
In PSI, the electron joins Chl a where it is re-energized (having lost energy in the electron transfer chain). Once the electron is re-energized, is transported down another electron transport chain (shorter one compared to the one on Photosystem II) before reaching NADP+ reductase. In turn, this enzyme (NADP+ reductase) transfers the electron to NADP+ to produce NADPH which is then moved to the Calvin cycle.
* The light-dependent reactions also produce ATP (along with NADPH).
The Calvin cycle, which includes light-independent reactions is the phase in which electrons produced by NADPH are used to form sugars using carbon dioxide.
The Calvin cycle is divided into three main stages that include:
Fixation
This is the first stage of Calvin cycle where an enzyme known as RuBisCO (ribulose bisphosphate carboxylase) catalyzes the reaction between carbon dioxide and three molecules of RuBP (ribulose bisphosphate). This reactions produces two molecules of 3-phosphoglyceric acid.
* The reaction between one molecule of carbon dioxide and RuBP gives two molecules of the three-carbon compound (3-PGA).
Reduction
Reduction is the second stage of the cycle. Here, Products of light-dependent reactions (ATP and NADPH) provide electrons that are used to convert molecules of 3-PGA into glyceraldehyde 3-phosphate. Here, six molecules of ATP and NADPH produce six molecules of the three-carbon molecule (glyceraldehyde 3-phosphate).
Regeneration
This is the last stage of Calvin cycle where a single molecule of glyceraldehyde 3-phosphate is transported to the cytoplasm to form various components needed by the cell and thus the plant in general. The remaining molecules are used to regenerate RuBP.
Uses of Chlorophyll
Chlorophyll has been used as a form for medicinal purposes.
Currently, it's used for:
- Detoxification
- Wound healing
- Constipation
- Reduce colostomy odor
- Reduce bad breath
Return to learning about Chloroplasts and Chromoplasts
And Photosynthesis
As well as Meristem Cells and Mesophyll Cells
Return to Leaf Structure under the Microscope
Return from Chlorophyll to MicroscopeMaster home
References
Constantin A. Rebeiz. (2013). Chlorophyll Biosynthesis and Technological Applications.
Eduardo Jacob-Lopes, Leila Queiroz Zepka and Maria Isabel Queiroz. (2017). Chlorophyll. Google books.
Lars Olof Björn. (2007). Photobiology: The Science of Life and Light.
Martin Schliep, et al. (2013). Formyl group modification of chlorophyll a: a major evolutionary mechanism in oxygenic photosynthesis. Plant, Cell and Environment (2013) 36, 521–527.
Mathias O. Senge, et al. (2014). Chlorophylls, Symmetry, Chirality, and Photosynthesis. Symmetry 2014, 6, 781-843; doi:10.3390/sym6030781.
Smita Rastogi & U. N. Dwivedi. (2007). Biomolecules (Introduction, Structure and Functions) Porphyrin.
Links
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4542031/
https://www.researchgate.net/publication/319703059_Chlorophylls_Chemistry_and_Biological_Functions
Find out how to advertise on MicroscopeMaster!

![Overview of the Calvin Cycle pathway by Mike Jones [CC BY-SA 3.0 (https://creativecommons.org/licenses/by-sa/3.0)] Overview of the Calvin Cycle pathway by Mike Jones [CC BY-SA 3.0 (https://creativecommons.org/licenses/by-sa/3.0)]](https://www.microscopemaster.com/images/512px-Calvin-cycle4.png)




