Giardia
Classification, Cell Biology, Life Cycle and Microscopy
Overview
Giardia is a genus consisting of a number of flagellated species with similar morphological characteristics. Members of this genus can be found in various environments (water, soil etc) and are responsible for giardiasis (a diarrheal illness) in both human beings and other vertebrates.
Given that the parasite is largely spread through the consumption of contaminated food and water (in areas with poor sanitation) by infected fecal matter, the illness is more common in third world and developing nations, and particularly areas with poor sanitation.
According to various molecular studies, Giardia species, as compared to many other eukaryotes, are some of the most divergent eukaryotes with distinct characteristics.
As such, they have proven to be some of the best models for studying various important pathways of eukaryotic cells as well as learning more about new and unique molecular mechanisms of their life cycle.
While it's suggested that the genus consists of numerous species, only six species have been well described, these include:
- G. intestinalis (also known as Giardia lamblia, or Giardia duodenalis)
- G. ardeae
- G. agilis
- G. microti
- G. psittaci
- G. muris
Classification of Giardia
· Kingdom: Protista - Members are neither animals, fungi or plants. A majority of these organisms are single-celled with unique and very diverse characteristics.
· Subkingdom: Protozoa - falls under the kingdom Protista and consist of eukaryotic single-celled organisms that either exist as parasites or as free-living organisms. Giardia, which falls under this subkingdom, are parasites of human beings and various vertebrates.
· Phylum: Sarcomastigophora - The term Sarcomastigophora is derived from the combination of the terms "Sarcodina" and "Mastigophora". Members of this phylum, including Giardia, are characterized as a locomotory organ (flagella in the case of giardia) as well as one type of nucleus. See Metamonada.
Whereas some members are free-living organisms, others like Giardia exist as parasites.
· Subphylum: Mastigophora - This division consists of about 1,500 flagellates that can be found in marine and freshwater environments. However, some species can be found in terrestrial environments in one form or another.
While most members of this division use flagella for swimming, some produce pseudopodia that allow them to trap food material. Both free-living and parasitic species fall under this subphylum.
· Class: Zoomastigophora - Members of this class (both free-living and parasitic) are classified by a body that is spherical and elongated (pear-shaped) as well as a central nucleus. A majority of species here also possess a single or several flagella for locomotion.
· Order: Diplomonadida - Members of this order are characterized by one or more flagella as well as being binucleated. A majority of the species are parasitic and thus depend on given hosts for their survival. As such, they are also found in anaerobic (or microaerophilic) environments.
· Family: Hexamitidae - For the most part, members of this family are intestinal parasites that parasitize both vertebrates and invertebrate hosts. Like Giardia, members of this family are also characterized by two nuclei as well as a body that is bilaterally symmetrical.
Host Specificity and Morphological Differences of Giardia Species
While Giardia species have a number of similar morphological characteristics, they can be differentiated based on a number of structural differences and the hosts they infect.
· Giardia agilis - Typically found in Amphibian hosts, G. agilis is characterized by a long and slender median body.
· Giardia muris - Often affect rodents and are characterized by a short and rounded median body.
· Giardia lamblia - Found in human beings and a number of other mammals, is characterized by a pear-shaped body and claw-shaped median bodies. Given that they are responsible for giardiasis in human beings, G. lamblia is one of the most common species.
· Giardia ardeae - Typically affect herons (coastal birds), are like Giardia lamblia as they are characterized by a pear-shaped body as well as a caudal flagellum that is similar to that of Giardia muris.
· Giardia psittaci - Affect a variety of birds and have similar morphological features as G. lamblia. However, they lack a marginal groove and an incomplete ventrolateral flange that is used for attachment purposes in G. lamblia.
· Giardia microti - Mostly affects voles and muskrats and is characterized by cysts that contain two trophozoites as well as mature ventral disks. However, when viewed under the light microscope, they possess morphological features similar to those of G. lamblia.
* While Giardia duodenalis (G. lamblia or G. intestinalis) is one of the most common species in the genus, molecular/genetic studies have found that it is not simply a uniform species but rather complex that consists of a number of genetically and phenotypically different genotypes.
These genotypes affect different hosts and include G. duodenalis (genotype A), G. enterica (genotype B), G. canis (genotype C), G. bovis (genotype E), G. cati (genotype F), G. simondi (genotype G).
Cell Biology
Based on morphological studies, Giardia species have been shown to present simple structural organizations when compared to other eukaryotic cells.
Following morphological studies on G. duodenalis, the organism was found to lack a number of organelles commonly found in other eukaryotic cells including mitochondria, peroxisomes as well as a well organized golgi complex.
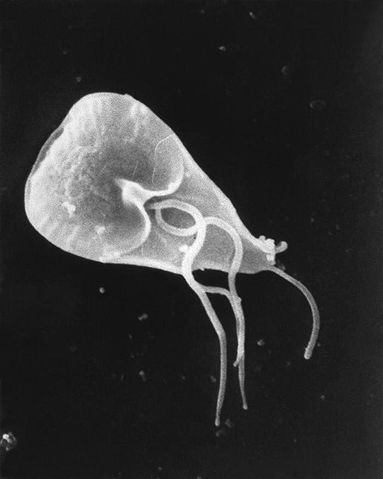
Generally, however, Giardia (trophozoite stage) is characterized by a pear-shaped body structure (a broad anterior and attenuated posterior). They range from 12 to 15um in length and between 5 and 9um in width.
One of the other defining morphological characteristics of Giardia is its unique cytoskeleton. The cytoskeleton consists of a median body that may be connected to the plasma membrane, caudal flagella or adhesive disc.
As flagellates, Giardia also possesses four pairs of flagella located at different parts of the body; anterior, posterior, caudal and ventral flagella.
As members of the order Diplomonadida, Giardia are binucleated which means that they have two nuclei (without nucleoli) at the anterior part of the body that are symmetrically positioned with respect to the symmetrical body structure of the organism.
At the anterior ventral side of the organism is also a ventral adhesive disk that is used for attachment to the mucosa.
* The medium body found in Giardia is composed of microtubules that have been suggested to play a role in supporting the posterior part of the organism.
Some of the other organelles found in the cytoplasm of Giardia include:
Life Cycle
The life cycle of Giardia consists of two main stages namely, the cyst stage and the trophozoite stage. Given that the cells go through the dormant cyst stage and the active/motile trophozoite stage, the whole life cycle can be said to go through two main processes that include the excystation and encystation stage.
Excystation
Basically, excystation refers to the stage in the life cycle of the organisms where trophozoite(s) are released from the tough outer covering known as the cyst. The infective stage of the parasite (the cyst) has to be ingested by the appropriate host first.
For the most part, this involves the consumption of contaminated foods or water. In the case of G. intestinalis, the cyst wall is made up of 60 percent carbohydrate and 40 percent protein. This composition produces a hardy wall that protects the parasite from hypotonic conditions in the environment that would otherwise cause lysis and destroy the cell.
Following ingestion by the host (human beings, mammals) the excystation process is activated by the stomach acids, ultimately causing the cyst to rupture in the small intestine.
Based on microscopic studies, flagella are the first structures to appear followed by the excyzoite body of the organism. While the host's stomach actively promotes the excystation process, the cysteine proteases in the peripheral vesicles have also been suggested to contribute to the degradation of the hardy cyst wall from within.
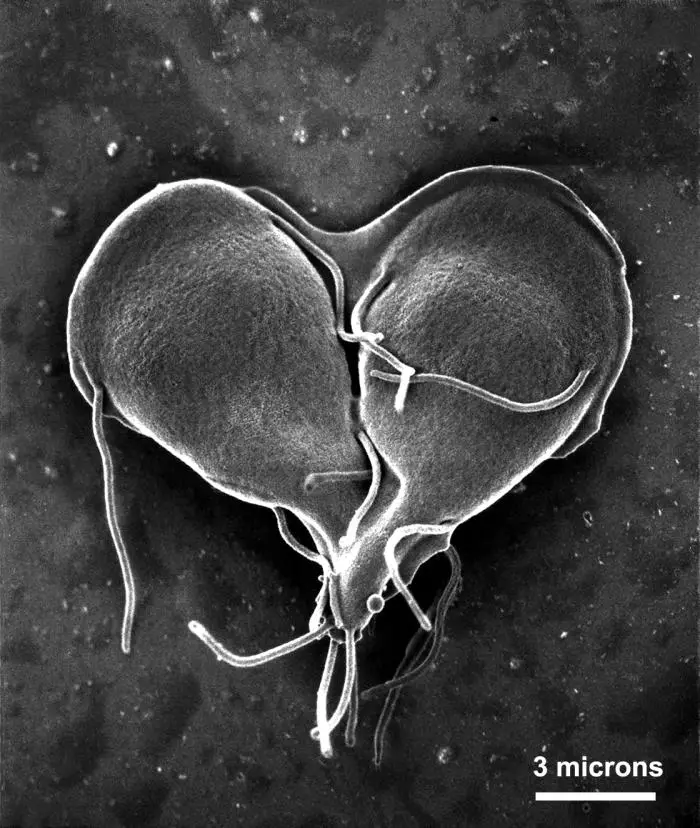
Once the excyzoite has been liberated, it goes through two phases of cytokinesis to form four trophozoites.
During this phase (division), a number of processes/activities also take place, these include:
- Increased metabolism
- Increased gene expression
- Segregation of organelles
- Upregulation of proteins involved in motility and adhesive discs
Once the trophozoite is fully developed/matured, it's characterized by the morphological traits mentioned above. Whereas the four pairs of flagella (12 to 15um in length) allow for the parasites to move, attach and dislocate from surfaces, adhesive discs function as suction cups that allow the parasites to bind to the surfaces.
Both the flagella and adhesive disc are made up of a and b tubulins as well as giardin proteins. Although the median body was initially thought to have no known function, it's now suggested to provide structural support.
* Flagella and the adhesive disc allows the parasite to remain attached to the intestinal walls even during peristaltic elimination.
Encystation
As compared to excystation, encystation is the process through which the active and motile trophozoites are transformed into non-motile cysts (infective state). Whereas excystation is completed when the parasite reaches the less acidic conditions in the small intestine, encystation is stimulated by high bile levels as well as basic pH.
This process starts with the internalization of the flagella as well as the fragmentation of the adhesive disc. As a result, the parasite is unable to remain attached to the intestinal walls.
A thick cyst wall is then formed encapsulating the excyzoite. Enclosed within the cyst wall are four nuclei of the excyzoite, microtubule structures, and flagella. Given that the cyst bodies are unable to attach to the intestinal walls, they are excreted along with feces.
* Due to the combination of carbohydrate chains, sugars, and proteins that make up the cyst wall, it's able to remain insoluble in aquatic environments.
Characteristics of Giardia cysts:
- ovoid in shape
- range between 10 and 15um in length
- cyst wall is about 0.3um in thickness
- contain less compact microtubules
* It only takes as few as 10 cysts to cause an infection.
* Giardia cysts can survive for several months outside the body of the host.
* The hardy cyst wall protects the parasite from such unfavorable environmental conditions as UV light, chlorine, extremely low temperatures.
Reproduction
In their life cycle, Giardia species have been shown to reproduce through asexual reproduction. Here, the trophozoites reproduce through simple binary fission (longitudinal binary fission).
This is characterized by nuclei division followed by division of the cytoplasm. This restores the binucleated state with each daughter cell containing a copy of each nuclei. While Giardia had been considered strictly asexual for a long time, emerging findings suggest that sexual reproduction exists among these parasites.
Giardiasis
As already mentioned, giardiasis is an infection of the small intestine caused by Giardia lamblia.
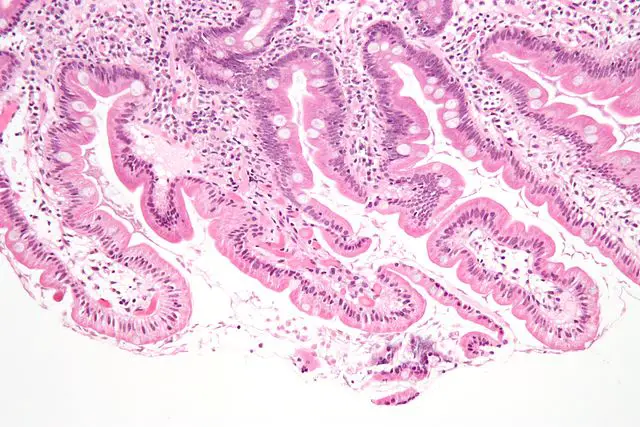
While the parasite invades the small intestine (particularly duodenum), studies have shown that it does not penetrate cells of the small intestine. Moreover, it does not invade the epithelium nor venture into the bloodstream.
With regards to the mechanism through which Giardia causes giardiasis, a combination of malabsorption and hypersecretion have been shown to be the primary causes of diarrhea.
By attaching to the cells of the epithelial wall/mucosa (enterocytes), Giardia was shown to cause shorting or the microvilli and thus lose epithelial wall function (absorptive function of the epithelial surface). As a result, the epithelial surface is incapable of properly absorbing such nutrients as glucose as well as water and other nutrients.
Apart from malabsoption, giardia infection was also associated with hypersecretion of chloride, which has been shown to contribute to fluid accumulation in the lumen of the intestine.
* Giardia species have been shown to share a number of metabolic and genetic characteristics with bacteria thus contributing to their unique characteristics that distinguish them from other eukaryotes.
Giardia has been shown to contain bacterial glycolytic enzymes that allow them to obtain energy through fermentation (by relying on pyrophosphate).
Microscopy
A number of techniques can be used to observe and study the morphological characteristics of Giardia species, these include:
Direct Wet Mount
Requirements
- Bright-field compound microscope
- Sample (this may involve the use of a stool sample)
- Glass slides
- Coverslips
- A wire loop/stick
- Saline
Procedure
· Using a dropper, place a drop of saline onto the center of the sterile glass slide
· Using a sterile wire-loop, obtain a small quantity of the sample (about 2mg) and add it onto the drop of water
· Mix the sample on the drop of saline
· Cover with a coverslip view under the microscope starting with low power
* A direct smear (wet mount) procedure helps preserve the parasite so that their motility is not lost. As a result, live parasites can be viewed under the microscope.
Observation
If cysts are present, they will appear ovoid in shape and colorless.
Trophozoites are not common from stool samples. However, in a case where a sample with trophozoites is used, they will also appear transparent but can be seen swimming in around.
* Staining may involve the use of iodine. While this makes it easier to identify the cysts, it also kills the organisms.
See Microscope Slide Techniques
Methylene Blue Staining
For this approach, 1 percent methylene blue solution and methanol are required
Procedure
· Using a sterile wire-loop, obtain a small quantity of the sample and make a smear at the central part of the glass slide
· Add a few drops of methanol to fix and allow to air dry
· Add a few drops of the stain and stand for 2 minutes
· Wash with tap water and allow to air dry
· Observe the slide under oil immersion
Observation
When viewed under the microscope, Giardia trophozoites can be seen clearly given that they take the color of the stain (if a giardia trophozoite positive sample was used). Unlike the case with direct wet mount, trophozoites, in this case, are immotile given that the preparation procedure kills live parasites.
* Giardia cysts are not visible because they do not take up the color of the stain.
Direct Immunofluorescence Assay (DFA)
Giardia can also be studied using Direct Immunofluorescence Assay. This approach involves tagging antibodies with fluorescent markers before adding them to a stool sample. The sample is then incubated before being visualized under the fluorescent microscope.
When viewed under the microscope, the parasite (cyst) is clearly visible as green in color and glowing.
See page on Cilia and Flagella
Return to Eukaryotes main page
Return to Parasites under the Microscope
Return from Giardia to MicroscopeMaster home
References
Andre G Buret. (2007). Mechanisms of epithelial dysfunction in giardiasis. ncbi.
Jian Han and Lesley J. Collins. (2012). Reconstruction of Sugar Metabolic Pathways of Giardia lamblia. International Journal of Proteomics
Volume 2012, Article ID 980829, 9 pages.
Johan Ankarklev et al. (2010). Behind the smile: cell biology and
disease mechanisms of Giardia species. © 2010 Macmillan Publishers Limited. All rights reserved.
Monali N. Rajurkar et al. (2012). A Simple Method for Demonstrating the Giardia Lamblia Trophozoite. ncbi.
Rodney D. Adam. (2001). Biology of Giardia lamblia. ncbi.
Links
https://www.ncbi.nlm.nih.gov/pubmed/9504342
https://www.cdc.gov/dpdx/resources/pdf/benchAids/Giardia_benchaid.pdf
Find out how to advertise on MicroscopeMaster!