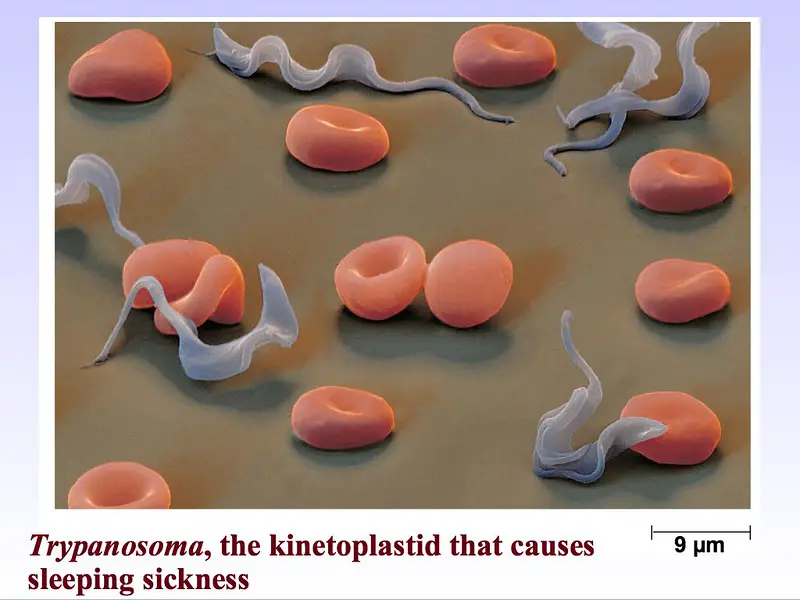
Kinetoplastids
Definition, Characteristics, Reproduction
Definition: What are kinetoplastids?
Classified under the phylum Euglenozoa, Kinetoplastids are flagellated protozoa classified under two monophyletic groups, namely, biflagellate bodonids and uniflagellate trypanosomatids based on their morphological traits.
Members of this group are distinguished from other protozoa by a number of unique structural components including the presence of a kinetoplast as well as kDNA within granule in their mitochondria. For the most part, Kinetoplastids are obligatory parasites with Trypanosoma and Leishmania species being responsible for human diseases.
Some of the kinetoplastids responsible for human disease include:
- Trypanosoma cruzi - causes Chagas disease
- Trypanosoma brucei - causes sleeping sickness
- Leishmania spp - responsible for leishmaniasis
* Some members of this class (Kinetoplastida) have been shown to exist as free-living flagellates (e.g. Bodo) while others are plant parasites (e.g. Phytomonas). In marine and terrestrial environments, free-living kinetoplastids feed on bacteria.
General Classification
· Kingdom: Protista - Eukaryotic organisms (single-celled and some multicellular organisms) that are not classified as plants, animals, or fungi.
· Phylum: Euglenozoa - Protists that exhibit one or more flagella in their motile (trophozoite) stage.
· Class: Kinetoplastida - Kinetoplastids is a class of the phylum Euglenozoa and is also referred to as Kinetoplastea.
· The subclass of class Kinetoplastida consists of Prokinetoplastina and Metakinetoplastida while the order consists of Prokinetoplastida, Trypanosomatida, Neobodonida, Eubodonida as well as Parabodonida.
Like kinetoplastids, Euglenids (Euglenoids) also fall under the phylum Euglenozoa. Unlike kinetoplastids, Euglenoids are either autotrophic or heterotrophic. As such, some species are capable of synthesizing their own food (Photosynthetic euglenoids) while others prey on bacteria and other small organisms in their environment (in marine and freshwater environments).
Reproduction and Life Cycle of Kinetoplastids
As compared to the free-living kinetoplastid protozoan (as well as plant parasite kinetoplastids), human kinetoplastid protozoan parasites complete their life cycle in invertebrate and vertebrate hosts. Depending on the organism, different morphological forms can be differentiated in each stage of their life cycle.
This section will focus on the general life cycle of Trypanosoma brucei and Leishmania spp as representatives of the group:
Reproduction and Life Cycle of Trypanosoma cruzi
T. cruzi parasites complete their life cycle by cycling between insects (Reduviid insects known as kissing bugs or assassin bugs) (invertebrate host) and the vertebrate host (e.g. human). In the sylvatic cycle, also known as the wild cycle, the parasite completes its life cycle by cycling between wildlife vertebrates and invertebrates (triatomine insects/kissing bugs).
During the life cycle of T. cruzi, the invertebrate (insect) acquires the trypomastigotes when they feed on the blood of an infected vertebrate. Once they are ingested, these parasites undergo further development (taking between 2 and 4 weeks) before they migrate into the hindgut of the insect.
Here, the forms that successfully migrate to the hindgut undergo transformation to the infective metacyclic trypomastigotes that are released into the environment along with fecal matter.
In the event that the insect defecates on the vertebrate host while feeding, these forms of the parasites can easily enter the body especially when the host scratches the wounded site (wound caused by the bug). However, this infection may also occur when human beings consume food material contaminated by fecal matter of the insect. In the invertebrate, these forms (metacyclic trypomastigote forms) differentiate to form trypomastigotes that enter the bloodstream.
* During feeding, the accumulation of blood into the gut of the insect (invertebrate host) forces accumulated excreta to be eliminated on the skin of the host. In the event that the host scratches this site, parasitic forms in the feces gain entry into the wound.
* The parasite can also gain entry into the body by using histolytic enzymes to break the skin and penetrate through active movement.
* Some of the parasites develop in the salivary gland of the insect and never migrate to the gut. As a result, they are transmitted to the vertebrate host through saliva of the insect as it feeds on blood.
Here, it's also worth noting that once they are ingested by the insect, a majority of the trypomastigotes are broken down by digestive enzymes of the insect. However, those that survive differentiate to form spheromastigotes that tend to be spherical in shape.
To avoid being eliminated from the body of the insect before they complete their development, epimastigotes that migrate to the gut attach onto the perimicrovillar membranes as they continue dividing to form the highly infective forms known as metacyclic trypomastigotes.
Once they enter the skin, the metacyclic trypomastigote forms penetrate the cells in this site and transform into amastigotes (replicative forms of the parasite) before transforming back to trypomastigote in the bloodstream.
* In the bloodstream of the vertebrate host, trypomastigote forms of the parasite divide asexually (through binary fission) as they increase in number.
* According to recent studies, T. cruzi are capable of reproducing sexually under specific circumstances.
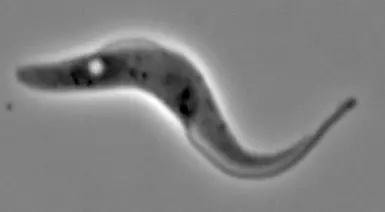
Trypanosome insect form cell
A live insect "procyclic" form trypanosome. Imaged by settling onto a glass slide and snapping a phase contrast image using a leica microscope.
The flagellar pocket (the "hole" near the left of the cell) is much more prominant in the insect form than in the bloodstream form (see image in same album).
During the life cycle of T. cruzi, the parasite exhibits different morphological forms with different structural characteristics:
· Trypomastigote - Elongated cell characterized by a kinetoplast that is located at the posterior part of the cell. The flagellum, which originates from the posterior of the cell spans along the entire length of the cell with the free part emerging at the anterior part of the parasite.
· Epimastigote - Elongated cells characterized by a centrally located kinetoplast. In this form of the parasite, the flagellum originates from the middle of the cell but ultimately emerges from the anterior part of the parasite.
· Promatogote - In this form of the parasite, the kinetoplast is at the anterior part of the cell. The flagellum also originates and emerges from this part of the cell.
· Amastigote - As compared to the other forms, the amastigote is spherical in shape. The kinetoplast appears as a dark body near the central section of the cell while the flagellum does not emerge from the cell body.
Reproduction and Life Cycle of Leishmania spp.
The life cycle of Leishmania species such as Leishmania amazonensis is similar to that of Trypanosoma species in that the parasite completes its life cycle between two hosts (a vertebrate (e.g. human) and invertebrate (female sandfly) host). During the life cycle of the parasite, the female sandfly is infected when it feeds on the blood of an infected person.
Here, the insect ingests amastigote forms of the parasite that transform into procyclic promastigotes. These forms undergo multiplication in the mid-gut of the insect before they migrate towards the anterior mid-gut where they continue dividing.
Here, the parasite again transforms to form the infective metacyclic promastigotes that are released into the skin of the vertebrate host through the insect's proboscis.
In the skin, the promastigotes invade macrophages and undergo transformation to form amastigotes. In the host cells, these new forms form and attach to the parasitophorous vacuole where they undergo intense multiplication ultimately causing the cell to burst.
Repeated infection of macrophages by amastigotes allows the parasite to proliferate. The cycle continues when the insect bites and feeds on the blood of the infected individual.
* While reproduction has for a long been thought to occur through binary fission only (a type of asexual reproduction), emerging studies have identified sexual reproduction (nuclear fusion) between amastigote forms that reside within cells of the host.
For this reason, reproduction in Leishmania is both asexual (binary fission where single cells divide to form two daughter cells) and asexual (nuclear fusion where male and female cells exchange genetic material).
Characteristics of Kinetoplastids
As previously mentioned, kinetoplastids are divided into two major monophyletic groups based on their morphology (biflagellate bodonids and uniflagellate trypanosomatids). Based on molecular studies, however, bodonids have been shown to be more diverse in nature.
Morphologically, all kinetoplastids are characterized by the presence of a kinetoplast which contains kDNA within the mitochondria. The general size of this sub-cellular structure varies from one organism/species to another. Whereas the kinetoplast measures about 0.6um in diameter in T. brucel, it is about 1um in T. cruzi.
Apart from the kinetoplast, kinetoplastids are also characterized by the presence of a flagellum that may be attached to the cell body in some cell. In cases where the flagellum is attached to the body, an undulating membrane is formed and plays an important role in motility.
Using given components of the cell membrane, such kinetoplastids as the African trypoanosomes and Leishmania parasites have also been shown to be capable of evading immune response of their host. This is particularly beneficial for the parasite given that it allows them to survive when they penetrate the skin of their vertebrate hosts.
Whereas Leishmania species depend on such surface molecules as lipophosphoglycan and protease gp63 to achieve this, trypanosomes evade immune response through the switching of their major variant surface glycoprotein.
Return to learning about Kinetoplasts
Return to understanding Parasites under the Microscope
Return to Parasitology main page
Return from Kinetoplastids to MicroscopeMaster home
References
Filardy, A. A. et al. (2018). Human Kinetoplastid Protozoan Infections: Where Are We Going Next?
Ken Stuart, Reto Brun, Simon Croft, Alan Fairlamb, Ricardo E. Gürtler, Jim McKerrow, Steve Reed, and Rick Tarleton. (2008). Kinetoplastids: related protozoan pathogens, different diseases.
Kenechukwu C. Onyekwelu. (2019). Life Cycle of Trypanosoma cruzi in the Invertebrate and the Vertebrate Hosts.
Teixeira, D. E. et al. (2013). The Cell Biology of Leishmania: How to Teach Using Animations.
Links
https://www.sciencedirect.com/topics/immunology-and-microbiology/kinetoplastida
https://www.tulane.edu/~wiser/protozoology/notes/kinet.html
https://www.intechopen.com/books/leishmaniases-as-re-emerging-diseases/immune-evasion-strategies
Find out how to advertise on MicroscopeMaster!