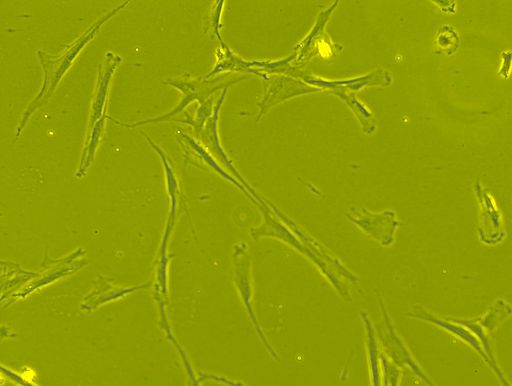
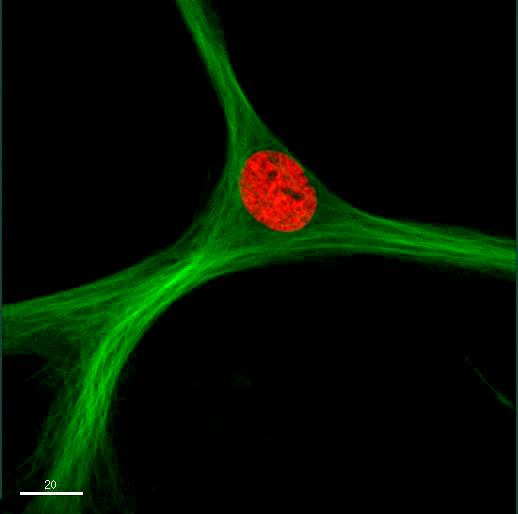
Mesenchymal Stem Cells
** Definition, Isolation Protocol/Differentiation
Definition
Essentially, mesenchymal stem cells are multipotent stem cells commonly found in the bone marrow. However, they can also be found in the umbilical cord blood, adipose tissue, and liver as well as the stroma of several other organs.
As multipotent cells in adults, these stem cells are not only capable of renewing themselves, but also giving rise to a number of other specialized cells that perform specific functions.
Because of their ability to differentiate into different types of specialized cells in the body, many researchers have focused their attention on using them for regenerative medicine.
Differentiation of mesenchymal stem cells can give rise to the following types of cells:
- Cells of the connective tissue - e.g. adipocytes and dermal cells
- Osteoblasts - bone cells
- Cartilage cells - e.g. chondrocytes
- Fibroblasts - e.g. tenocytes/tendon cells
- Striated muscle cells - e.g. skeletal muscle cells
- Myoblasts
* In the bone marrow, mesenchymal stem cells make up about 0.01 of the total nucleated cells (1 in 10,000).
Following fertilization of the egg cell, stem cells known as embryonic stem cells makes up the inner mass of the embryo. Like stem cells, these cells are undifferentiated. However, they are pluripotent and can differentiate and give rise to derivatives of the primary germ layers.
As the embryo develops and stem cells differentiate to form various function cells, studies have shown the switch from embryonic stem cells to post-embryonic results in loss of potency. This means that the new stem cells that remain fixed in given body tissues and organs can only give rise to certain types of cells.
In addition to a loss of potency, these cells are very few in number and only make a very small percentage of cells wherever they are located. Mesenchymal stem cells are a good example of adult stem cells.
Bone Marrow-derived Mesenchymal Stem Cells
Like hematopoietic multipotent stem cells, mesenchymal stem cells can also be found in the bone marrow. Also known as bone marrow stromal cells, these cells are located within the medullary stroma.
According to the International Society for Cellular Therapy, adults’ stem cells in the human bone marrow have to meet a number of criteria in order to be accepted as mesenchymal cells.
These include:
· Have to be plastic-adherent - When mesenchymal cells were first identified in 1970, they were shown to adhere to culture plates
· Must be able to express a number of accessory receptors - These include CD105, CD73, and CD90
· Must be able to differentiate into osteoblasts, chondroblasts, and adipocytes (in vitro)
In a study to investigate some of the differences between peripheral blood mesenchymal stem cells and bone marrow mesenchymal stem cells in mice, results showed that both have the same cell surface marker expression.
As compared to the mesenchymal stem cells found in blood, those in the bone marrow have a higher proliferative capacity. Moreover, they have a greater capacity to produce osteoblasts and chondrocytes.
Like the other adult stem cells, bone marrow-derived mesenchymal stem cells are multipotent and therefore capable of differentiating into several types of cells under certain conditions. Thus far, they have been shown to be capable of becoming endothelial cells, osteoblasts, smooth and cardiac muscle cells, adipocytes, tendon cells, as well as fibroblasts.
Adipose-derived Mesenchymal Stem Cells
Also known as lipocytes/fat cells, adipocytes are a type of cells that store fats and make up the adipose tissue. Like the bone marrow, the adipose tissue also contains mesenchymal stem cells.
These stem cells are easier to obtain given that they can simply be isolated from a fat tissue biopsy. This is an especially simpler process compared to isolation of bone marrow-derived mesenchymal stem cells which involves aspiration of the bone marrow.
They are also similar to bone marrow-derived mesenchymal stem cells in that they are multipotent and thus capable of giving rise to a number of specialized cells. They are also capable of self-renewal and therefore continue to renew themselves in the adipose tissue.
According to the stem cell committee of Internal Society for Cellular Therapy (Mesenchymal and Tissue Stem Cell Committee of the International Society for Cellular Therapy), cells can only be considered adipose-derived mesenchymal stem cells if they meet the following criteria:
- Plastic adherence in standard culture conditions
- Should be able to differentiate to adipocytes, osteoplasts, and chondrocytes
- Should be able to express the following surface markers: CD105, CD73, and CD90
These stem cells are also metabolically active and have also been shown to play an important role in the revascularization of damaged tissue, inhibiting apoptosis as well as immunomodulation. This is achieved through the secretion of extracellular matrix factor as well as a variety of other substances including cytokines and growth factors.
Differentiation
Mesenchymal stem cells have the ability to divide and renew themselves. In addition, they, being multipotent cells, can differentiate to produce a range of cells.
Generally, the differentiation of mesenchymal stem cells to produce various mature and function cells is divided into two main parts that include lineage commitment and maturation.
During the lineage commitment phase, the mesenchymal stem cells divide to produce a committed progenitor. As well, the maturation phase results in the production of mature phenotypes (functional cells) from the progenitor.
Based on in vitro studies, the type of cells produced through the differentiation process is dependent on a number of factors. For instance, before they start to differentiate, mesenchymal stem cells exhibit a fibroblast-like morphology.
Following their interaction with an elastic extracellular matrix, they not only cause deformation of the matrix but also become more rounded. This has been shown to influence adipogenesis.
A matrix that is stiffer is able to withstand the force produced by these cells and also influences the spreading of these cells. In the process, this has been shown to influence the osteogenic lineage.
By influencing the shape of these cells as well as their capacity to spread, geological constraints have a direct influence on lineage commitment.
Some of the other factors that influence the differentiation of mesenchymal stem cells include:
Transforming growth factor-beta (TGFβ) and Bone morphogenetic proteins (BMPs) - Individually, at different concentrations, subtypes of TGFβ1 (TGFβ1, TGFβ2, and TGFβ3 and BMPs) have been shown to influence the differentiation of mesenchymal stem cells into different functional cells.
Whereas BMP4 can influence the differentiation to mesenchymal stem cells along the adipogenic lineage, BMP2 and rosiglitazone work together to influence this differentiation.
In the TGFβ/BMPs signaling pathway, the TGFβ/BMPs regulate adipogenic and osteogenic differentiation of the mesenchymal stem cells through the activation of the canonical Smad-dependent pathways as well as the non-canonical Smad-independent signaling pathway.
Wnt (wingless-type) / β-Catenin Signaling - The Wnt family consists of glycoproteins involved in various functions. With regard to the differentiation of mesenchymal stem cells, the protein has been shown to act through its interaction with β-catenin.
Here, activation of the Wnt signaling pathway promotes the osteogenic differentiation while inhibiting the differentiation of mesenchymal stem cells in the adipogenic lineage.
Hedgehogs - Based on a number of studies, various components of the Hedgehog signaling pathway have been shown to be highly expressed in mesenchymal stem cells.
During the differentiation of mesenchymal stem cells towards the adipogenic lineage, studies have shown there to be a downregulation of the Hedgehog signaling as well as a result of reduced expression of Gli proteins.
The activation of this signaling was shown to block this differentiation while a combination of the Hedgehog and BMP signal was associated with osteogenic differentiation. This is evidence that the Hedgehog proteins influence the lineage of differentiation.
Notch - Like the Hedgehog proteins, Notch proteins are also involved in mesenchymal stem cell differentiation. This has been demonstrated through Notch signaling blockage.
Studies have shown this to result in the autophagy-mediated differentiation of mesenchymal stem cells along the adipogenic lineage through the PTEN-PI3K/AKT/mTOR pathway.
By inhibiting Wnt/β-catenin signaling, Notch signaling has been shown to suppress osteogenic signaling.
Some of the other factors that influence the differentiation of mesenchymal stem cells include:
- Fibroblast growth factors (FGSs)
- Age
- Oxygen concentration
- Supplementary factors
Isolation Protocol
As mentioned, mesenchymal stem cells can be isolated from a number of sources in the body including adipose tissue, the bone marrow, as well as cord blood, etc. For each of these sources, different methods are used for the isolation of mesenchymal stem cells.
The following are some of the methods used to isolate mesenchymal stem cells from cord blood, the adipose tissue, and bone marrow:
Isolation of Mesenchymal Stem Cells from Cord Blood
To isolate mesenchymal stem cells from cord blood, the following material are required:
- Cord blood
- Phosphate buffered saline
- RPMI medium (RPMI 1640) - Growth medium
- Gradient media (e.g. Ficoll-Paque Premium)
Procedure
· Cord blood is first diluted with the growth medium at the ratio of 3:1 - Three (3) parts of the blood and one part medium
· Density gradient centrifugation (at 400 times gravity) is then used to isolate mononuclear cells - This is conducted at room temperature for about 30 minutes
· Using another centrifuge tube, the mononuclear cells are mixed with Phosphate-buffered saline at the ratio of 1:3 (three parts Phosphate-buffered saline and one part mononuclear cells)
· The mixture is centrifuged for 10 minutes at 400 times gravity/400xg (at room temperature)
· The supernatant is carefully removed and the cells resuspended by adding culture medium
· Lastly, contents are incubated overnight at 37 degrees C and 5 percent of carbon dioxide
Isolation of Mesenchymal Stem Cells from the Adipose Tissue
Requirements
Some of the requirements used for the isolation of mesenchymal stem cells from adipose tissue include:
- Ammonium chloride (NH4Cl)
- Blood sample consisting of adipose tissue (Blood/saline solution)
- 40 percent of dulbecco's modified eagle medium
- Well plates
- PBS (Phosphate buffered saline)
- Nylon mesh - 100um
- Nylon mesh - 40um
- Collagenase
Procedure
· Using a micropipette, carefully aspirate off the saline and oil phases
· Using the Phosphate buffered saline, wash the fat about 3 times for about 5 minutes and then discard the low phase
· Add collagenase to the remaining content and incubate at 37 degrees C for between 1 and 4 hours
· Add about 10 percent of fetal bovine serum to neutralize the collagenase and then centrifuge the fat for about 10 minutes (at 800xg)
· Using a micropipette, aspirate the floating adipocytes and lipids so that you retain a pellet of stromal vascular fraction
· Using 160mM ammonium chloride, resuspend the stromal vascular fraction and then incubate for about 10 minutes. This is followed by centrifugation of the contents for about 10 minutes at 400xg
· Layer the cells on the Histopaque gradient and again centrifuge for 30 minutes at 1000xg (1000 times gravity)
· Use PBS (phosphate-buffered saline), resuspend the pellet, and then filter the cells using the 100uM mesh and again pass the cells through the 400uM
· Again, centrifuge the contents for about 10 minutes at 400xg before resuspending the pellet in 40 percent FBS/DMEM
· Plate the cells and incubate overnight at 37 degrees C and 5 percent of carbon dioxide
Isolation of Mesenchymal Stem Cells from the Bone Marrow
Requirements
- Properly prepared mice hind legs
- Growth culture medium
- Syringe
- Incubator
Procedure
As compared to the other sources, isolation of mesenchymal stem cells from the bone marrow tends to be more intrusive.
The following steps are used to isolate these cells from mice:
· To obtain bone marrow from the bone, the flushing method is used - This involves flushing the bone marrow from the bone using a syringe that contains about 1ml of the growth medium (growth culture media)
· Bone marrow stromal cells are suspended in 5ml of the medium (the medium also consists of 20 percent FBA as well as 1 percent ampicillin)
· Cells are then seeded in tissue culture plates before being incubated for 24 hours at 37 degrees C
· Non-adherent cells are removed and the adherent cells maintained in growth culture media for 24 hours - This is often repeated several times a week with the media being changed for each growth cycle
· Cells are passaged in suspension culture
· Lastly, immunocytochemistry analysis can be used for analysis of the cells
Treatment
Unlike totipotent cells, mesenchymal stem cells only give rise to a given number of functional cells and are therefore classified as multipotent stem cells.
The fact that they can give rise to several types of cells is beneficial in that they can be used for treatment purposes, especially in regenerative therapy.
Based on a number of studies, mesenchymal stem cells have been shown to have the potential to treat a number of pathologies including diabetes, cartilage disease, cancer, and cardiac ischemia, etc.
Here, the potential of these cells to treat various diseases is attributed to several important properties that include the ability to migrate as well as the capacity of these cells to produce various mediators.
As already mentioned, the interaction between mesenchymal stem cells and their surroundings not only influences the differentiation lineage but also how they spread. In the event of an injury, these cells have been shown to migrate to the damaged tissue and start differentiating depending on the local conditions.
For this reason, it's possible to introduce them to parts of the body affected by given pathologies so that they can differentiate and produce desired cells. As well, they have been shown to produce a range of bioactive mediators including cytokines, anti-inflammatory substances as well as growth factors that are involved in the treatment of some pathologies.
In some studies involving mice, for instance, the use of bone marrow-derived mesenchymal stem cells has been shown to reduce infarction as well as the loss of gray-white matter.
Following migration to the damaged tissue, a process that is stimulated by various mechanisms and factors, mesenchymal stem cells contribute to regeneration and tissue by activating various mechanisms.
As mentioned, they are able to secrete a range of mediators (e.g. anti-inflammatory and angiogenic agents, etc) in response to signals associated with injuries and thereby contribute to regeneration/tissue repair.
Contact with some immune cells has been shown to influence immunosuppressive effects. This is particularly important in the case of bone marrow transplant etc as it enhances immune tolerance which in turn allows foreign cells to proliferate.
With regards to cancer treatment, the interaction between these cells and cancer cells has also been shown to affect further tumor development.
* Although mesenchymal stem cells are already being used for regenerative medicine in some spheres, it's worth noting that studies on the use of these cells for the treatment of other pathologies is ongoing.
Advantages
Some of the main advantages of mesenchymal stem cells include:
· They can renew themselves - This is a big advantage given that self-renewal ensures that they are never depleted. As such, they can continue giving rise to a variety of functional cells when required.
· Differentiation - As already mentioned, mesenchymal stem cells continue to give rise to a variety of functional cells in adults. In medicine, this is particularly beneficial as there are no ethical issues when it comes to using stem cells obtained from adults. For this reason, they can be isolated when needed for medical trials among other studies.
· Lastly, they have the potential to treat a variety of diseases including cancer which gives hope to many patients across the world. With continued studies and medical trials, it's expected that a breakthrough will see mesenchymal stem cells being used for treatment purposes.
See also: Hematopoietic Progenitor Cells, Exosomes and Stem Cells
Return from Mesenchymal Stem Cells to MicroscopeMaster home
References
Anny W. Robert, Bruna H. Marcon, Bruno Dallagiovanna and Patrícia Shigunov. (2020). Adipogenesis, Osteogenesis, and Chondrogenesis of Human Mesenchymal Stem/Stromal Cells: A Comparative Transcriptome Approach.
Bruno Machado Bertassoli et al. (2013). Mesenchymal stem cells - emphasis in adipose tissue.
Lina Badimona, Blanca Oñatea, and Gemma Vilahur. (2015). Adipose-derived Mesenchymal Stem Cells and Their Reparative Potential in Ischemic Heart Disease.
Q Chen et al. (2016). Fate decision of mesenchymal stem cells: adipocytes or osteoblasts?
Links
https://www.sciencedirect.com/topics/medicine-and-dentistry/mesenchymal-stem-cell
Find out how to advertise on MicroscopeMaster!