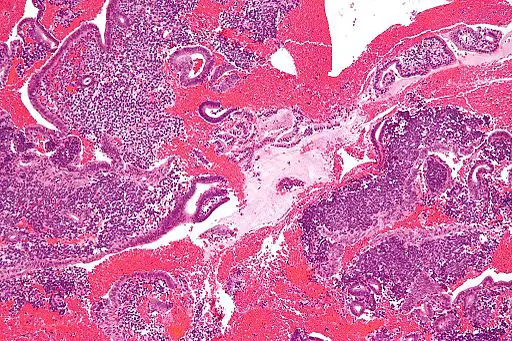
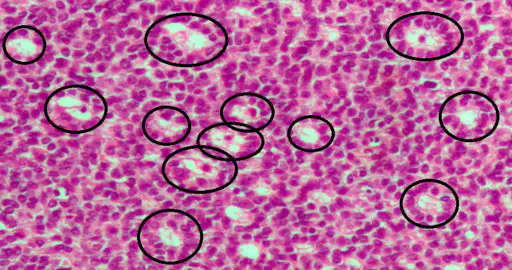
Stromal Cells
** Definition, Function, Examples, Vs Stem Cells & Cancer
Definition
Essentially, stromal cells are a diverse type (heterogeneous) of potent cells commonly found in the bone marrow. However, they have also been isolated from several other parts of the body including the adipose tissue, umbilical cord blood, skin, and amniotic fluid, etc.
While they are also referred to as mesenchymal stem cells in some literature, it's worth noting that mesenchymal stem cells are themselves a type of stromal cells. Regardless, they are capable of differentiating, both in vivo and in vitro, to give rise to several types of specialized cells (e.g. adipocytes).
This, in addition to plastic adherence and the expression of given cell surface markers, are some of the characteristics associated with these cells. In the body, stromal cells play a number of important roles ranging from angiogenesis to promoting hematopoiesis.
Some of the surface markers expressed on stromal cells include:
- CD44
- CD29
- CD45
- CD105
- CD90
Examples
There are several types of stromal cells in the body.
Mesenchymal stem cells - Also known as multipotent stromal cells, mesenchymal progenitor cells, and mesenchymal stromal cells, mesenchymal stem cells are some of the most common stromal cells.
While they are commonly found and normally isolated from the bone marrow, mesenchymal stem cells can also be found in the adipose tissue, the umbilical cord tissue, as well as the placenta. There are a number of characteristics associated with these cells.
These include:
- Spindle-shaped
- Exhibit adhesion properties (plastic-adherent cells). As such, they can adhere to tissue culture plastic or the surface of microcarriers etc.
- Differentiate in vitro - They can differentiate to give rise to various types of cells in culture
- Capable of self-renewal - This may occur through symmetrical and asymmetrical division
Pericytes - Initially, pericytes were thought to be a type of progenitor of mesenchymal stem cells. However, different types of studies have shown the two types of cells to have a number of important differences that sets them apart.
Generally, pericytes are commonly found in the basement membrane of capillaries, thus protecting the endothelial cells, as well as the postcapillary venules.
However, they have been shown to share part of their origin with mesenchymal stem cells. Like mesenchymal stem cells, pericytes are multipotent cells and can differentiate to give rise to different types of cells (e.g. osteocytes and neural cells, etc.) when activated.
Unlike mesenchymal stem cells, pericytes are slow expanding cells in culture and are also classified as contractile cells.
While pericytes from different vessels share several characteristics (e.g. the ability to differentiate), they have also been shown to exhibit various differences (heterogeneity).
Fibroblasts - Fibroblasts are some of the most abundant stromal cells in the body. Like mesenchymal stem cells, fibroblasts are spindle-shaped cells with a flattened oval nucleus and thin cytoplasmic processes.
As cells of the connective tissue, these cells can be found in various body organs including the uterus, skin, and gastrointestinal tract, etc. They are characterized by a number of markers including CD90, collagen type I, and fibroblast activation protein among others.
Apart from morphology, fibroblasts are also identified from their role in ECM (extracellular matrix) secretion. Following activation, fibroblasts differentiate to produce myofibroblasts.
* Myofibroblasts are also described as stromal cells. They play an important role in epithelial homeostasis.
Fibroblast-like stromal cells - Fibroblast-like stromal cells are cells that can be found in a number of regions including the foreskin and ovary. In the ovary, these cells can be found in the cortex or the medulla.
Here, they have been associated with the production of collagen (collagen 1 and collagen 111) as well as the production of steroids.
Functions
Differentiation
As mentioned, stromal cells can differentiate to give rise to different types of cells.
Here, some of the processes involved include:
Chondrogenesis
Chondrogenesis refers to the process through which chondrocytes are produced through the differentiation of mesenchymal stem cells (chondrocytes are a type of cells that form the cartilage). As is the case with stem cells, chondrogenesis involves two main phases that include proliferation and differentiation of the mesenchymal stem cells.
Once they are activated (e.g. by the presence of transcription factor sox-9), mesenchymal stem cells undergo proliferation to increase in number. The cells then undergo differentiation to produce chondrocyte progenitors known as chondroblasts.
In turn, chondroblasts are responsible for the production/secretion of the matrix that not only separates the cells, but also promotes their transformation into chondrocytes.
Unlike chondroblasts, chondrocytes, which are now surrounded by small empty pockets/spaces within the matrix, known as lacuna, are less active (low metabolism and slowed growth) due to the thick matrix which slows the diffusion of oxygen and other substances required for cellular activities.
* Due to the very slow rate at which the cartilage is repaired following damage, researchers have been working on ways to use mesenchymal stem cells for treatment purposes by stimulating their proliferation to produce chondroblasts for repair.
Osteogenesis
The term osteogenesis refers to bone formation. Following birth, bone formation may involve bone growth (increase in size and one grows), bone remodeling as well as fracture healing. In a fetus, the process of bone formation starts with mesenchymal cells (in the mesenchyme) creating an ossification center and undergo differentiation to form osteoblasts.
Like chondroblasts, these cells are highly active and have a secretory role. In this case, osteoblasts are involved in the secretion in the extracellular matrix which consists of a number of contents including collagen fiber. In the extracellular matrix, osteoblasts undergo differentiation to produce osteocytes which are contained in lacunae.
This is followed by calcification of the bone matrix before the outer portion of the bone starts forming. Here, the mesenchymal cells are replaced by the fibrous periosteum as blood vessels continue developing within the spongy part of the bone.
Adipogenesis
As the name suggests, adipogenesis is the process through which adipocytes are produced through the differentiation of mesenchymal stromal cells.
Following activation of the mesenchymal stromal cells (by peroximse proliferator-activated receptor y), these cells commit to the adipoblast lineage and undergo a series of differentiation to produce adipocytes. This is an important process that results in the development of the adipose tissue.
* Compared to other stem cells and progenitor cells in the body, mesenchymal stromal cells have been shown to be easy to isolate. Moreover, they exhibit a high rate of proliferation, easy expansion in culture, and also lack HLA class II receptors.
For this reason, they are ideal for transplantation especially for the purposes of repairing damaged tissues and organs.
Role of Stromal Cells in the Immune System
In the body (in vivo), mesenchymal stromal cells have been shown to influence the activities of adaptive and innate immune cells. For this reason, they are said to have an immunomodulatory effect. Here, the manner in which mesenchymal stromal cells influence the immune system is associated with their general traits.
Generally, these stromal cells have been shown to have low immunogenicity. For this reason, they do not provoke immune responses against themselves. This is largely due to the fact that unlike some foreign invaders, mesenchymal stromal cells express low levels of HLA class 1 (human leukocyte antigen class 1) but do not express HLA-DR.
* Cells that lack HLA class 1 can be easily identified and destroyed by natural killer cells. For this reason, expressing low levels of this molecule is important because it protects these stromal cells from the actions of natural killer cells
Learn more about immunology here.
Like some of the immune cells, mesenchymal stromal cells have receptors (chemokine) that allow them to migrate to sites of inflammation. At the site of inflammation, they have been shown to associate with different types of immune cells to mitigate the regeneration of damaged tissue.
During the regeneration process, mesenchymal stem cells are suspected to bind to immune cells (in the same way programmed death-ligand I bind to immune cells) resulting in reduced or lost immune function.
Here, these stromal cells have also been shown to affect the production of cytokines by immune cells which in turn prevents other immune cells (e.g. macrophages and regulatory T cells) from being activated and mobilized to the affected site.
In doing so, mesenchymal stromal cells are able to not only regulate the actions of immune cells, but also contribute to tissue regeneration at the affected site.
Given that some of the immune cells (e.g. macrophages) play an important role in destroying damaged/necrotic cells, the immunomodulatory effects of mesenchymal stromal cells are important given that they help establish homeostasis that allows for tissue regeneration and repair as well as immune functions at the site of injury or infected site.
* Because of their ability of mesenchymal stromal cells to migrate and home damaged tissues, researchers have taken advantage of this property for the purposes of treating such conditions as ischemic cerebral disease, spinal cord injuries, as well as myocardial infarction etc.
Here, the cells are introduced into the body through intravenous injections and migrate to the affected site where they are involved in tissue regeneration.
Some of the other mechanisms through which mesenchymal stromal cells modulate immune responses include:
· Inhibit or minimize the proliferation and development of T cells - This is achieved by preventing expression of cyclin D2 and production of nitric oxide
· Regulate activation, differentiation, and division of B cells - This is achieved by contributing to the arrest of cell cycle at the G0/G1 phase as well as releasing molecules like Blimp-1
Bone Marrow Stromal Cells
In the bone marrow, stromal cells are generally referred to as non-hematopoietic stem cells. This is because they do not differentiate to produce blood cells. However, they have been shown to regulate the process (hematopoiesis) by regulating hematopoietic stem cells. One of the ways through which stromal cells are thought to influence hematopoietic stem cells is through the osteolineage path.
As already mentioned, some of the stromal cells give rise to osteoblasts during bone formation. Here, the osteoblast lineage cells have been shown to produce several cytokines that regulate hematopoietic stem cells. Some of these cytokines include thrombopoetin, granulocyte colony-stimulating factor, and CXCL12 (a CXC chemokine).
On the other hand, while enforced expression of parathyroid hormone receptor 1 results in an increase of hematopoietic stem cells with an expansion of osteolineage cells, a reduction of these cells has been linked to a reduction of hematopoietic stem cells within the bone marrow.
Apart from the cytokines mentioned above, N-Catherin, a protein that is expressed on some osteolineage cells, is also suspected to influence hematopoietic stem cells. Here, however, it's worth noting that it is still debatable as to whether the protein has a significant impact on hematopoietic stem cells and more studies are required.
Some of the other important functions of stromal cells include:
Formation and maintenance of blood vessels - The loss of pericytes in blood vessels has been shown to result in hyperdilation and conditions like edema and embryonic lethality. These findings are evidence of the significance of pericytes in the formation and maintenance of blood vessels. Specifically, these cells (also known as perivascular cells) can be found surrounding the vascular tube.
Regulating blood flow - Because of their contractile property, pericytes contribute to the vasodilation and vasoconstriction of capillaries
Lymphoid organogenesis - In association with lymphoid cells, stromal cells have been shown to play an important role in the organogenesis of lymphoid tissue
Support parenchymal cells – As cells of the connective tissues, stromal cells also play an important role in supporting parenchymal cells (functional cells)
Stromal Cells Vs Stem Cells
Stromal cells and stem cells have a number of similarities despite the fact that they are different types of cells.
Some of the similarities between the two types of cells include:
They can be found in both adults and fetal tissues - While stromal cells (mesenchymal stromal cells) were first identified in the bone marrow, they, like stem cells, are now known to be present in fetal tissues. Both types of cells can also be found in adults. Stem cells in adults are known as adult stem cells.
Potency - Like adult stem cells, stromal cells are multipotent cells and thus capable of giving rise to different cell types. For instance, in adults, these cells are capable of differentiating to produce fat cells, cartilage cells, as well as bone cells.
Pericytes, which are also a type of stromal cell, can give rise to astrocytes, oligodendrocytes, and neurons. In children and adults, stem cells, which are also multipotent cells, can be found in such tissues as the bone marrow and can differentiate to produce a number of cell types.
Self-renewal - One of the main characteristics of stem cells is their capacity for self-renewal. Generally, this means that they are able to divide and produce more stem cells. This characteristic has also been identified in stromal cells like mesenchymal stromal cells.
For this reason, mesenchymal stem cells are sometimes referred to as "true stem cells"
* While mesenchymal stem cells (mesenchymal stromal cells) are sometimes referred to as stem cells, they are not true stem cells given that they are pre-differentiated and only capable of giving rise to given cell types of cells.
As such, they are different when compared to pluripotent cells (common the embryo and fetus) which can be stimulated to produce any cell type.
Stromal Cells and Cancer
Essentially, cancer is the term used to refer to diseases that are characterized by abnormal cell growth. Here, the cells divide uncontrollably thus affecting the normal functions of cells and tissues.
In tumor microenvironments, tumor cells have been shown to recruit stromal cells, among other cells, thus contributing to progression of the condition. Some of these cells include pericytes, endothelial cells, bone marrow mesenchymal/stromal cells, as well as fibroblasts.
As previously mentioned, stromal cells can migrate to an injured site where they contribute to tissue regeneration during repair. With regards to cancer, however, studies have shown some tumor cells to recruit these cells to the tumor microenvironment where they are gradually transformed into tumor-associated stromal cells.
As compared to normal stromal cells, tumor-associated stromal cells produce higher levels of proteins, other signaling molecules as well as tumorigenic factors that promote development of these conditions.
Return from Stromal Cells to MicroscopeMaster home
References
Gabriele Bergers and Steven Song. (2005). The role of pericytes in blood-vessel formation and maintenance.
Hisatsugu Goto and Yasuhiko Nishioka. (2018). Fibrocytes: A Novel Stromal Cells to Regulate Resistance to Anti-Angiogenic Therapy and Cancer Progression.
Karen M. Bussard et al. (2016). Tumor-associated stromal cells as key contributors to the tumor microenvironment.
William Cathery, Ashton Faulkner, Davide Maselli, and Paolo Madeddu. (2018). Concise Review: The Regenerative Journey of Pericytes Toward Clinical Translation.
Links
https://www.ptglab.com/news/blog/are-mesenchymal-stem-cells-mscs-true-stem-cells/
https://www.researchgate.net/publication/275114964_Mesenchymal_Stromal_Cells
Find out how to advertise on MicroscopeMaster!