What are Cytokines?
Types, Receptors, Functions - In Inflammation
Defintion: What are Cytokines?
Generally, cytokines are a variety of regulatory/signaling molecules (small proteins or glycoprotein) produced by various nucleated cells in the body. As products of nucleated cells (various cells of the immune system), they play a crucial role in the regulation of various aspects of immunity including the intensity and duration of immune responses by immune cells, etc.
Based on studies, cytokines have also been shown to be pleiotropic and therefore have varying impacts on different types of cells. Different types can also influence the activities of others and vice versa.
In addition to mediating and regulating immune responses, cytokines are also involved in the production and development of all the different types of blood cells, through a process known as haematopoiesis, angiogenesis as well as tumorigenesis among a few other important functions.
* Because of their role in body immunity, cytokines are also being used more as diagnostic and therapeutic agents.
Types of Cytokines
- Interferons
- Chemokines
- Interleukins (many interleukins are considered to be lymphokines)
- TNF (tumor necrosis factor)
Functions
Interleukins
Produced by such leukocytes as lymphocytes and monocytes (and a number of other cells in the body e.g. ) interleukins are glycoproteins involved in the activation and differentiation of immune cells. In addition, they also play an important role in the proliferation, migration, maturation, pro and anti-inflammatory activities, as well as adhesion of these cells.
Together with interleukin receptors, interleukins belong to a superfamily (IL superfamily) that is made up of proteins. While it's easy to generalize the functions of these cytokines, this is largely dependent on the type of interleukins. Currently, over 43 members of this superfamily have been identified (IL-1 to IL-43).
Like some of the other cytokines, interleukins are made up of proteins. Typically, this is in response to invading pathogens/antigens. While high volumes may be produced depending on the type of interleukins and the invading organism, a small quantity of the molecule is required to activate biological effects.
While a good number of interleukins elicit an action on the same cells that produced them, some can enter the bloodstream which allows them to be transported and elicit biological effects on distant cells in the body.
While IL-2 elicits biological effects on T cells (which produced them), IL-1 can enter the bloodstream and reach the central nervous system.
Some of the other properties of interleukins include:
- Synthesis is a self-limited process
- Stimulate up-regulatory and down-regulatory mechanisms
- Have redundant functions in the body - Given interleukins can cause different effects on various types of cells
- Can influence the synthesis and functioning of other interleukins
As already mentioned, different types of interleukins have different functions.
The following are some of the functions of a few of these molecules:
· IL-1 - Indirectly stimulates immune responses via various effector proteins and other cytokines.
· IL-2 - Interleukin 2 plays an important role in growth regulation of T cells.
· IL-3 - Stimulates the production of myeloid progenitor cells by hematopoietic stem cells. However, an interaction between IL-3 and IL-7 results in the production of lymphoid progenitor cells from hematopoietic stem cells.
· IL-4 - Interleukin 4 is involved in a number of biological processes ranging from the proliferation of T cells and B cell stimulation to humoral and adaptive immunity. Moreover, it contributes to the production of a number of cells including dendritic cells, Th1 cells as well as Interferon Gamma cells.
· IL-5 - Like Il-4 and IL-13, interleukin 5 is also involved in stimulating the growth of B-cells. However, it is also involved in increased secretion of immunoglobin as well as activation of eosinophils.
· IL-6 - In addition to being the primary mediator in such illnesses as fever, interleukin 6 has also been shown to overcome the blood-brain barrier in activating the expression of Prostaglandin E2 in the hypothalamus which results in body temperature change.
Some of the other common interleukins include:
- IL-8 - involved in the induction of chemotaxis
- IL-10 - involved in inflammation as well as regulation of the immune responses
- IL-18 - involved in both immune responses in the body (innate and adaptive responses)
- IL-33 - involved in immune responses of Th2 (e.g. parasitic infections)
Interferons (IFNs)
Interferon is a family consisting of widely expressed signaling proteins. Like the other cytokines, interferons are also released by cells of the host's immune system in response to such invading organisms as bacteria and viruses.
They are also released to respond to tumorous cells in some organisms. Currently, three types of interferons have been identified. These include Type I IFNs, Type II IFN, and Type III IFNs.
Type I IFNs (Type I Interferons)
Type I interferons are divided into two major groups that include IFN-α and IFN-β as well as a number of additional isotypes that include, among others, IFN-κ, IFN-ω, and IFN-δ. While only one type of IFN-β exists, IFN-α is further divided into several subtypes including IFN-α1, IFN-α2, IFN-α3, IFN-α4, IFN-α5, and IFN-α6 among others.
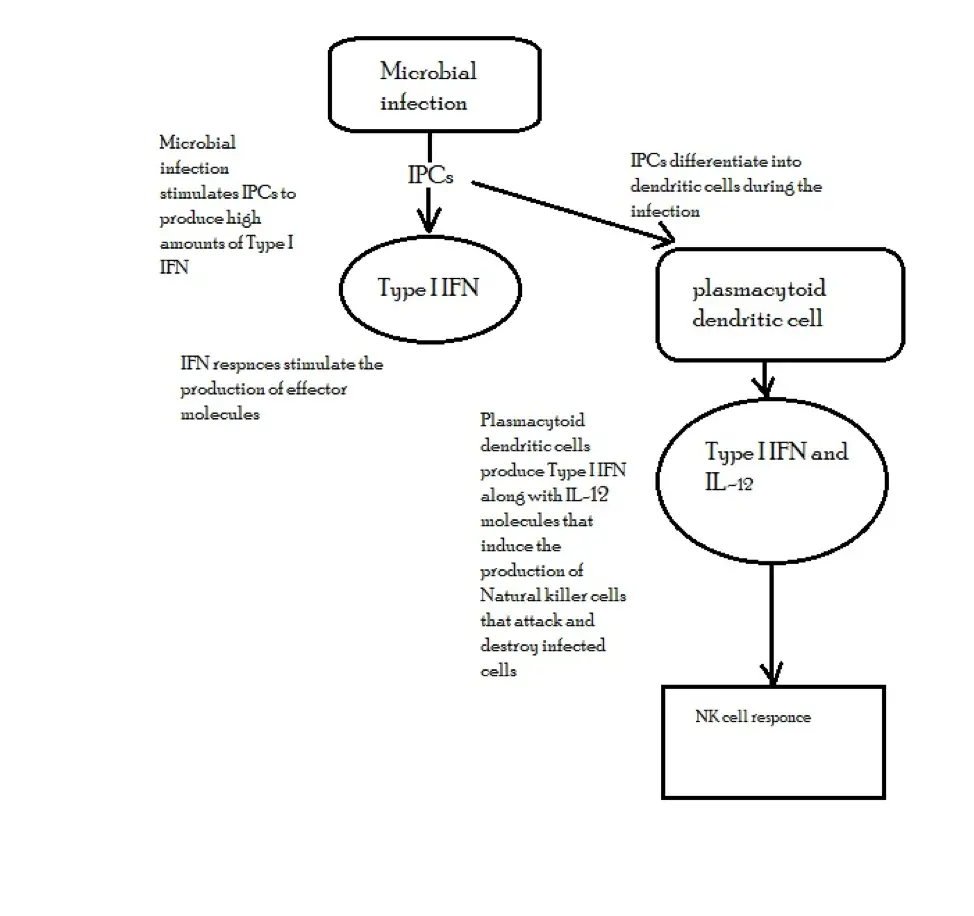
In the body, the production of Type I IFNs is dependent on the presence of various microorganisms. For instance, following a viral infection, a signaling pathway that causes phosphorylation, dimerization as well as passage of the interferon response factor 3 (IRF3) to the nucleus is activated.
Along with a number of other transcription factors, IRF3 activates that synthesis of IFN-β gene that binds to the interferon receptors located on the surface of an infected cell which ultimately results in interferon response.
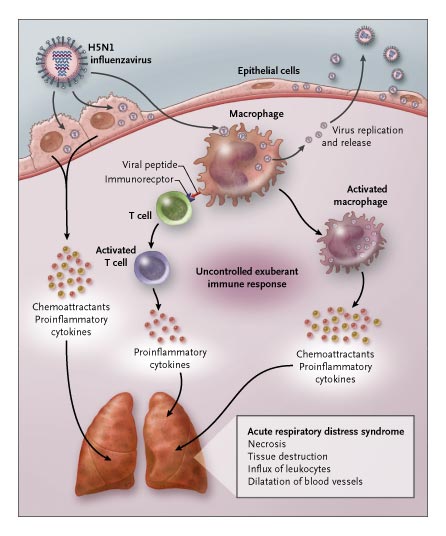
Through these responses, interferons help in the recruitment of effector molecules that protect the cells against infections (bacterial and viral infections).
By activating the production of natural killer cells and macrophages, interferons contribute to the destruction of both the viruses and infected cells.
* Cells responsible for the production of IFN-α and IFN-β are collectively known as interferon-producing cells (IPCs) or natural interferon-producing cells.
IFN production may be presented as follows:
Functions of Type I interferons (IFN-α and IFN-β) are generally divided into three main categories that include:
· Influence resistance to viral replication in cells - This is achieved through the destruction of viral mRNA required for viral replication as well as inhibiting the translation of viral proteins.
· Promote ligand increase - Type I interferons promote an increase in ligands to the receptors of NK cells which in turn stimulates them to attack and lyse infected cells.
· Activate the destruction of infected cells by NK cells and macrophages.
* Given that Type 1 interferons also plan a role in immunosuppressive activities, they are also used for the purposes of treating autoimmune diseases.
Type II Interferon
Type II interferon is made up of a single cytokine known as IFN-y. This cytokine is largely produced by THI T cells, activated natural killer cells as well as CD8+ T cells.
Unlike cytokines of Type I interferons, the gene responsible for encoding the Type II cytokine is located in chromosome 12 in human beings.
Moreover, IFN-y has been shown to be different from the other interferons in that it does not produce a potent antiviral effect. Rather, it largely serves to activate effector cells.
While it's produced by T cells in adaptive immunity (following an increase in antigen), IFN-y is produced by natural killer cells in the innate immune system and acts as a mediator. In the adaptive immune system, increased production of IFN-y is promoted by IL-12 and IL-18.
On the other hand, IL-4 and IL-10 correspond to the negative regulators involved in its production. Apart from T cells, B cells and professional antigen-presenting cells have also been shown to play a role in the production of IFN-y.
Like cytokines of Type I interferons, IFN-y also contributes to cell response to viral infections. For instance, by activating and causing the induction of MHC (major histocompatibility complex), IFN-y has been shown to play a role in long-term control of viral infections in cells. In the process, it also coordinates the transition from innate to adaptive immunity.
Some of the other functions of IFN-y include:
· Macrophage activation - By promoting the activation of macrophages, IFN-y contributes to phagocytic and pinocytic activities of these cells and thus contributes to microbial destruction.
· Inhibiting cell growth - In the body, IFN-y has also been shown to inhibit cell growth and thus promote apoptosis.
Type III interferons
Unlike Type II interferon that only consists of a single cytokine, Type III is divided into three important cytokines that include IFN-λ1, IFN-λ2, and IFN-λ3. Also known as IL-28 (a and b) and IL-29, Type III interferons are characterized by a structure that is more similar to proteins of the family IL-10.
As well, the signaling pathway of these cytokines has been shown to resemble that of Type I interferons in that they are dependent on the actions of IRFs and NF-kB.
While these cytokines also regulate a number of similar functions as Type I interferons, they primarily function in mucosal epithelial cells, as well as liver cells in human beings, where they serve to protect them from viral infections.
Chemokines
Chemokines are a type of protein cytokine that play an important role in chemotaxis. As such, there may be signals that guide certain immune cells to the affected site.
Currently, about 50 chemokines have been identified. They are divided into four families based on the location of N-terminal cysteine residues in their three-dimensional structure.
These include:
CC chemokines - CC chemokines are characterized by two adjacent cysteines (known as CC chemokine ligands) located near the N-terminus of the structure. Of the 27 CC chemokines (CCL-1 to CCL-28) that have been identified so far, only 6 have 6 cysteines while the rest are characterized by four of these molecules.
For the most part, CC chemokines are chemotactic for monocytes with a few being chemotactic for lymphocytes. As such, they promote the movement/migration of these cells to the affected site.
CXC chemokines - As compared to CC chemokines, CXC chemokines are generally characterized by two cysteines located at the N-terminus. Here, the cysteines are separated by an amino acid.
So far, seventeen of these cytokines have been identified. They are chemotactic for neutrophils and thus promote the movement/migration of these cells to the affected site.
CXCL8 (also known as IL-8) is one of the most common CXC chemokines. It's responsible for the recruitment of neutrophils as well as maintaining inflammatory reactions.
With regards to neutrophil recruitment, CXC chemokines have to interact with the appropriate receptors located on the surface of neutrophils.
By interacting with CXCRI and CXCR2 receptors located on the surface of neutrophils, ELR-positive CXC chemokines are able to recruit them to action.
C-chemokines - Currently, only two of CC chemokines have been identified. These include the XCL1 and XCL2 chemokines which are characterized by two cysteines, one of which is located at the N-terminus while the other is located downstream.
CX3C chemokines - Like the other chemokines, the CX3C chemokines also contain two cysteines at the N-terminus. However, the cysteines are separated by three amino acids. CX3CL1, which is the only chemokine in this group, is not only a chemoattractant, but also a cell adhesion molecule.
To serve its function, CXCL1 has to bind to CX3CR1, a receptor located on the cell that expresses it.
Chemokine receptors - These include the G-protein-coupled receptors (consisting of 7-transmembrane α-helical segments) and atypical receptors are expressed on all white cells involved in immunity.
While each type of the receptors can bind to different chemokines (within the same family), a single chemokine can also bind to several receptor subtypes. Regardless, according to studies, a majority of these chemokines have been shown to display receptor specificity.
While regulating leukocytes through the peripheral lymphoid tissues is one of the functions of chemokines, their primary role entails recruiting such cells as neutrophils and T cells to the site of inflammation. Here, the migration of the leukocytes to the affected site is achieved by the stimulation of actin filaments.
Some of the other important functions of chemokines include:
- Promoting angiogenesis (CXC chemokines)
- Wound healing (CXC chemokines)
- Development of various non-lymphoid organs
- Priming certain T cells
* For the most part chemokines are relatively small in size, ranging between 8 and 14 kDa.
* To achieve their functions, chemokines have to be released in large amounts so as to establish a concentration gradient necessary to influence cell migration.
Tumor Necrosis Factor
Tumor Necrosis Factor (TNF) consists of a group of proteins involved in a number of physiological and pathological processes. Currently, about 40 members of the superfamily (TNF) have been identified with TNF-α and TNF-β being the most notable examples.
Some of the other members of TNF include:
- CD40 ligand
- OX40 ligand
- FAS ligand
- GITR ligand
TNF-alpha (TNF-α)
Also known as TNF or TNFSF2, TNF-α is a multifunctional cytokine involved in such processes as apoptosis and coagulation among others.
In human beings, TNF-α genes are located on chromosome 6. This allows the cytokine to be expressed and produced by such immune cells as macrophages, monocytes and T cells in response to invading pathogens or the presence of cytokines like IFN-y.
Some of the main characteristics of TNF-α include:
· Following activation of macrophages and other cells, TNF-α is synthesized as a 26-kD nonglycosylated type II membrane protein.
· Two types of TNFα that include mTNFα and sTNFα.
· Due to adipose tissue expression, TNF-α is sometimes referred to as an adipostat.
Functions of TNF-α
As mentioned, TNF-α is a multifunctional cytokine that ranges from immune roles to programmed cell death.
Immune function - Like the other cytokines, TNF-α plays an important role in immunity. In particular, TNF-α attracts certain immune cells to the affected site by stimulating the expression of adhesion molecules by vascular endothelial cells.
This makes it possible for immune cells to adhere to blood vessel walls and successfully migrate to the infected site and destroy invading pathogens (bacteria and viruses).
Induces the production of chemokines that are involved in inflammatory responses - These cytokines guide immune cells to the affected site.
Apoptosis - TNF-α promotes the programmed cell death of tumor cells by promoting the recruitment of proteins involved in death signaling.
Biological functions - When produced in large amounts, TNF-α has been shown to induce reduced blood pressure or shock during such events as severe infections. In some cases, however, a high concentration of this cytokine results in low blood sugar concentration as well as intravascular thrombosis.
TNFβ - Also known as Lymphotoxin, TNFβ is a type II transmembrane protein. The expression of this cytokine is stimulated by activated lymphocytes. With regards to functions, TNFβ is a potent mediator involved in various immune and inflammatory responses that have similarities to those of TNF-α.
Like TNF-α, TNF-β is also involved in the following processes:
- Apoptosis
- Coagulation
- Cell proliferation and differentiation
Receptors
Generally, receptors are membrane-bound molecules on which cytokines bind in order to elicit given cell functions. Therefore, cytokines can be said to influence cell functions/activities by binding to specific receptors.
Based on structure and activities, receptors and their corresponding cytokines are divided into the following groups/families:
Type 1 cytokine receptors - This family includes receptors for a number of interleukin cytokines (IL-2 to IL-12) as well as a number of other cytokines (e.g. GM-CSF, LIF, and Epo, etc) and hormones (e.g. prolactin, growth hormone, etc).
These receptors are characterized by given conserved motifs located in their extracellular amino-acid domain. However, they lack an intrinsic protein tyrosine kinase activity.
Type II cytokine receptors - Members of this family are multimetric receptors characterized by heterologous subunits. They are, for the most part, receptors for interferon cytokines (e.g. IFN- alpha and beta).
Chemokine receptors - There are four types of chemokine receptors (corresponding to the four types of chemokines). They are typically described as G-protein-coupled receptors with a 7 transmembrane structure.
Tumor necrosis factor receptors (TNFR) - Members of this family are characterized by a cysteine-rich domain consisting of three disulfide bonds that surround a core motif.
TGF-beta receptors - These receptors include TGFBR1, TGFBR2, and TGFBR3 which are basically serine or threonine kinase receptors.
Return to Immunology main page
Return from Cytokines to MicroscopeMaster home
References
Andreas Wack, Ewa Terczyńska-Dyla & Rune Hartmann. (2015). Guarding the frontiers: the biology of type III interferons. Nature immunology. Arunabha Ray and Jagdish Joshi. (2016). Cytokines and their Role in Health and Disease: A Brief Overview. ResearchGate.
Justiz Vaillant and Ahmad Qurie. (2019). Interleukin. www.ncbi.nlm.nih.gov.
Vinicius L. Ferreira et al. (2017). Cytokines and Interferons: Types and Functions.
Wen-Ming Chu. (2014). Tumor necrosis factor. ncbi.
Links
https://www.sinobiological.com/what-is-tumor-necrosis-factor-tnf-a-6649.html
Find out how to advertise on MicroscopeMaster!
![3D medical animation still showing secretion of Cytokines by www.scientificanimations.com [CC BY-SA 4.0 (https://creativecommons.org/licenses/by-sa/4.0)] 3D medical animation still showing secretion of Cytokines by www.scientificanimations.com [CC BY-SA 4.0 (https://creativecommons.org/licenses/by-sa/4.0)]](https://www.microscopemaster.com/images/512px-Cytokine_release.jpg)