Microscopy Culture and Sensitivity
Urine, Sputum, Blood and Stool
Overview
In microbiology, culture techniques are used for the purposes of evaluating tissues and fluids. This makes it possible for the technician to study, if present, infectious processes in the sample.
Here, it's possible to identify microorganisms in the growth media, their characteristics or their impact on cells/tissue through microscopy.
In addition to propagating microorganisms in a medium, tests known as sensitivity (susceptibility) testing are used to determine the right treatment for the infection. Here, a range of drugs can be used in order to study their impact on the microorganism and thus determine whether they are the ideal treatment option for given infections.
Urine Microscopy Culture and Sensitivity
Although urine is expected to be sterile, a small number of bacteria can be found in the urine of healthy individuals. However, in the case of an infection, it's important to culture urine in order to not only determine the type of microorganism responsible for the infection, but also the ideal treatment option.
Here, the presence of bacteria or other microorganisms in urine may be an indication of an infected urethra, kidneys, uterus, or the urinary bladder.
Basic Principles of Urine Culture
While a normal urinalysis tests allows for the detection of various disorders in the urinary tract system, a urine culture is normally requested in the event of such symptoms as blood in the urine, backache, or a burning sensation during urination etc.
A culture test may be requested in cases where routine urinalysis detect some abnormalities (e.g. during routine urine tests for pregnant women) - This is required even if the patient is asymptomatic.
Collecting Urine for Culture
Based on various studies, urine has been shown to be an excellent growth medium for various organisms. For this reason, collecting urine for culture requires very special care in order to avoid contaminating the urine with other microorganisms that were not originally in the urine.
Some of the most important general considerations for urine collection include:
· A urine sample should be collected from the patient. Therefore, urine from such sources as the urinal or a bedpan should never be used
· The patient should be encouraged to wash the urethral/vaginal with soap and cleaning water before collection. This helps prevent microorganisms commonly found on the skin from contaminating the urine - Using a disinfectant is prohibited given that it may inhibit the growth of the microorganism in the event it's introduced into the urine sample.
· The patient/individual collecting the urine sample should always wash their hands with soap and clean water to avoid any contamination.
· If all the above requirements are met, a sterile container should be used to collect the urine sample - such containers are normally provided in a clinical setting and contain preservatives to preserve the urine sample before culture.
· First-morning urine is usually recommended given that it's not diluted.
· The urine should be collected midstream - In addition to washing the hands and parts of the urinary tract, this helps minimize the chances of contamination with normal skin flora.
* For some patients, collecting urine may involve inserting a catheter into their bladder. Here, it's still important that the patient cleans their urethral or vaginal areas in order to avoid contamination. The catheter is then inserted into the bladder of the patient to collect the urine.
* A urine sample should be cultured as soon as possible. However, in cases where this is not possible, it should be stored in a container with a preservative and refrigerated. Such samples can only be used within a period of 48 hours.
* Potentially infectious body fluids should always be handled with care.
Enhanced Quantitative Urine Culture (EQUC)
For urinary tract infections, studies have shown enhanced quantitative urine culture to be one of the most effective standard tests for the detection of microorganisms in the sample.
Unlike most of the older techniques, a higher volume of urine is used in EQUC to detect more bacteria. Here, the sample is incubated in an environment that contains a high concentration of carbon dioxide for a period of 48 hours in three different growth media.
Growth media used includes:
· Chocolate agar plates - The urine sample in this media is incubated in conditions consisting of 5 percent carbon dioxide concentration, 35 degrees C for a period of 48 hours.
· BAP (blood agar plate) - Culture is incubated aerobically at 35 degrees C for a period of 48 hours.
· MacConkey agars - The culture is also incubated in anaerobic conditions at 35 degrees C for a period of 48 hours.
* One of the biggest advantages of using this technique is that it allows for the detection of live microbes in various urine samples where standard culture methods may report no growth.
* Bacteria (or any other microorganisms that form colonies) can then be stained for microscopy procedure.
* A positive culture refers to a culture in which colonies of bacteria (or other microorganisms) grow.
Sensitivity
As mentioned, sensitivity tests are used to study the impacts of various antimicrobial agents on the microorganism. Here, the primary goal is to determine whether the antimicrobial agent prevents the organism from proliferating and how effective the agent is.
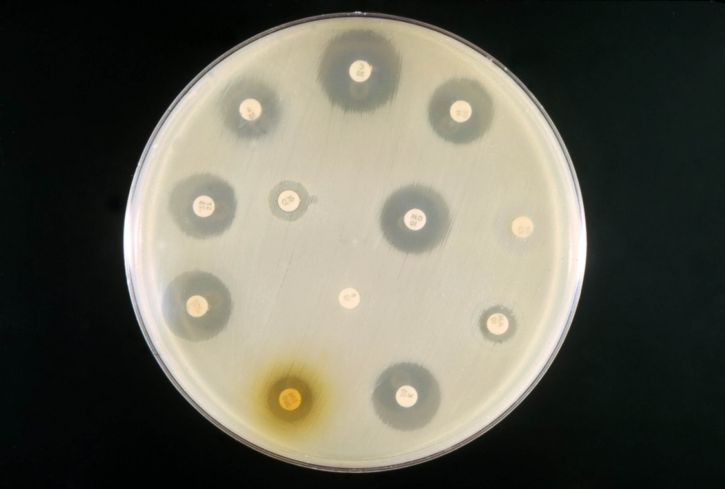
One of the most commonly used sensitivity tests is the disk diffusion test. While there are a number of other tests used for sensitivity, the disc diffusion test has become the standard because it's very simple and practical.
For this method, the bacterial inoculum is first placed on the surface of a Mueller-Hinton agar plate. Twelve paper antibiotic disks are then placed on the agar surface before being incubated for about 24 hours at 35 degrees C.
Following the incubation period, the plate is investigated for zones of growth inhibition around each of the disks. The diameter of these zones is related to susceptibility of the organism as well as diffusion rate of the drug.
Some of the antimicrobials used for this test include penicillin, narrow-spectrum cephalosporin, and anti-pseudomonal penicillins with β-lactamase inhibitors among others.
* Microscopy makes it possible to estimate the number of bacteria cells in urine. In healthy individuals, the number of bacteria present in urine is less than 10,000(104) per ml of the urine sample.
If a sample contains 1,000,000(105) bacteria cells per ml of blood, this is indicative of an infection that requires treatment.
Sputum Microscopy Culture and Sensitivity
Sputum can be described as a thick fluid that is produced in the lungs and respiratory tract. Given that a variety of organisms can cause respiratory infections, a sputum test is used for the purposes of detecting the organism (bacteria or fungi) and thus determine the most effective treatment.
Sputum Collection for Culture
As is the case with other body fluids required for culture, several important steps have to be taken into account to ensure that the sample is collected in the right way.
These include:
· Before collecting sputum, the patient should be informed that nasal secretions and saliva are not sputum
· The patient should rinse his/her mouth with a clean glass of water before sputum collection - This is done in order to remove any food particles that may be present
· Sputum should be collected in a sterile, disposable container with a cap - these containers should always be checked for any potential leakages
· To collect the sputum, patients are advised to take a deep breath. After a moment of holding the breath, they are then advised to cough deeply and release the sputum into the container - Container should always be labeled in order to avoid any confusion
· For patients who are unable to produce sputum, aerosol inhalation is often used to induce sputum production. Cotton swabs are often used for young patients
· If the sputum has to be transported, then it should be tightly capped to avoid leaking and refrigerated at a temperature range between 2 and 8 degrees C
Culture
Routine sputum culture is aimed at detecting the presence of such pathogens as Haemophilus influenzae, S. pneumoniae, yeast, and S. aureus among others. Typically, the set of culture media used includes blood agar, chocolate agar, and MacConkey agar.
The process involves the following steps:
· The purulent part of sputum should be washed in sterile saline - This is aimed at removing mucus and other contaminants that may be present in the sputum sample
· Before inoculating the sample, they are normally diluted in sterile saline (1: 10) and additionally in 10ul of aliquots
· The prepared sample of sputum is then inoculated on the agar plates (MacConkey agar, heated sheep blood agar, and chocolate agar)
· In the chocolate agar plate, an optochin disc is normally added for the identification of S. pneumoniae
· For the blood agar and chocolate agar plates, incubation is carried out at about 35 (to 37) degrees C in an atmosphere that contains elevated levels of carbon dioxide for 24 to 48 hours. Also, the MacConkey plate is incubated at 35-37 degrees C in air
Common results of routine culture include:
· Whitish colonies with rounded shape in the blood and chocolate agar - Indicates the presence of Candida albicans
· In MacConkey agar, colonies often indicate the presence of Pseudomonas, Acinetobacter, or Enterobacteriaceae which are not of significant clinical importance
· In blood and chocolate agar plates, grey-white dry colonies are indicative of B. catarrhalis - This can be confirmed by sugar degradation tests
· In blood and chocolate agar plates, water-drop colonies are indicative of H. influenzae. These colonies are often clear and larger in chocolate agar plates
· The presence of S. pneumoniae is normally characterized by clear colonies that are flat with a concave center and green zones. Here, a zone of inhibition is formed around the optochin disc
Sensitivity
A sensitivity test is normally performed when bacterial growth is significant.
Here, different approaches are used for different organisms:
· Disc-fussion method - This is often used for staphylococci and members of the family Enterobacteriaceae
· Mueller-Hinton agar - Used for S. pneumoniae. This is normally used to determine susceptibility of the organism to drugs like erythromycin and tetracycline etc
· Chocolate agar - Susceptibility tests of H. influenzae strains to drugs like tetracycline and chloramphenicol is performed on chocolate agar plates
Microscopy
Microscopic examination of sputum makes it possible to study morphological characteristics of microorganisms that are present in the sample. Here, Gram staining is performed to investigate the presence of Gram-positive or Gram-negative bacteria.
Possible results of microscopic examination include:
· Gram-negative diplococci - B. catarrhalis
· Gram-positive cells that look like yeast - Candida
· Clusters of Gram-positive cocci - S. aureus
· Tiny Gram-negative coccibacilli - H. influenzae
See more on Gram positive and Gram negative bacteria.
Blood Microscopy Culture and Sensitivity
The presence of bacteria in the blood is known as bacteremia. These organisms or their toxins can also poison blood resulting in septicemia. Apart from bacteria, a number of other microorganisms like yeast can also be found in blood resulting in infections like candidiasis.
These infections can result in septic shock, a fatal condition associated with life-threatening low blood pressure. Using a blood culture, it becomes possible to determine the type of microorganism in blood for proper treatment.
See article on performing a blood smear.
Culture
For blood culture, blood culture bottles are often used to collect blood (aerobic and an anaerobic bottle). Here, however, the volume/quantity of blood collected is dependent on the patient.
Adults - For adults, each of the bottles (aerobic and an anaerobic bottle) is inoculated with about 10ml of blood.
Pediatric - To volume/quantity of blood collected from children and infants is largely dependent on their body weight. Typically, an anaerobic bottle is used to collect blood apart from cases where an anaerobic infection has been detected.
Unlike bottles used to collect blood from children, these bottles used are specially formulated. The recommended blood-to-broth ratio is between 1:5 and 1:10.
* Before collecting blood, it's important to disinfect the bottle tops with about 70 percent of isopropyl alcohol and to clean the puncture site with alcohol and chlorhexidine. The site should also be allowed to dry before being punctured to collect blood.
* Inoculating the anaerobic first is always recommended in order to prevent air from entering the sample.
Important steps to consider before sample collection includes:
- Draw blood from the veins and not arteries
- Always check collection bottles for damage, contamination, breakages and the expiry date
- Do not collect blood from the venous/arterial catheters to avoid contamination
- Clearly label the collection bottles to avoid confusion
Once the blood has been collected for culture, it's normally incubated for about 3 days at 37 degrees C. Under these conditions, it's possible to recover more than 97 percent of microorganisms that are of clinical significance.
Results
False-positive - A false-positive result is a result in which an organism that was not present in the bloodstream of the patient is detected growing in the culture. Such results are a sign of contamination.
Negative and positive results (negative and positive blood cultures) - In the event that the suspected microorganism is detected in a culture, then this is a positive result. However, if it's absent, then the result is a negative one.
Sensitivity
To determine sensitivity of the microorganisms in blood culture, Kirby-Bauer diffusion method is used on Muller-Hinton agar. For Gram-positive bacteria, some of the drugs used include Ampicillin, Clindamycin, Erythromycin, and Clindamycin among others.
For Gram-negative bacteria, drugs used to test sensitivity include Ciprofloxacin, Tetracycline, Nalidixic acid, and Cefoxitin among others.
How do antibiotics kill bacteria?
Microscopy
Blood staining is necessary for microscopy. Some of the stains used to stain a blood smear include Wright's stain (nuclear and cytoplasmic characteristics of different types of cells and microorganisms in the blood), Giemsa stain (often used for diagnosis of protozoan and spirochete parasites in blood), and Romanowsky stains (used to stain the nuclei of blood parasites).
Stool Microscopy Culture and Sensitivity
A stool culture is often used to identify the type of microorganism responsible for an infection at the lower digestive tract. Given that there are naturally existing bacteria in the digestive tract (normal flora), this test is important in that it makes it possible to differentiate between normal flora and pathogenic microorganisms responsible for the infection.
Basic steps of stool sample collection:
· Before collecting a stool sample, patients are advised not to use antacids, and drugs used for indigestion, barium/bismuth, diarrhea medicine, or laxatives for about a week
· The stool sample should always be collected in a specimen container with a lid - This is often provided by the care provider
· A clean bedpan or commode can be used to collect a sample before a small sample is put in the collecting container (with a lid) - A tongue blade can be used for the transfer
· A pair of gloves should be used to prepare a stool sample collection
· Some collecting containers contain preservatives. Therefore, patients should be reminded not to pour it out
The container should be labeled appropriately to avoid confusion.
Stool Culture: Basic Principles
Before inoculation, the formed stool has to be prepared. This involves suspending a small sample in saline in order to make a suspension. However, in the case of watery diarrhea, preparing a suspension using saline is not necessary. Once the suspension has been prepared, inoculation of the agar plate is the next step.
Some of the media used include:
· MacConkey agar - This is a general-purpose plating medium that can be used for Shigella spp, Yersinia enterocolitica, and E. coli.
· Xylose-lysine-deoxycholate agar - This is a selective medium that has to be used in addition to the general-purpose media. Along with other media like deoxycholate citrate agar, this medium is used for the growth of a given organism.
· Mueller-Hinton agar with 5% sheep blood - This is also a selective media used for culture of Campylobacter spp.
· Alkaline bile salt agar (BSA) and thiosulfate citrate bile salts (TCBS) are some of the selective media used for culture of Vibrio strains. However, MacConkey agar can also be used.
* Incubation requirements vary between different organisms. Whereas Vibrios are incubated at 35 to 37 degrees C overnight, Campylobacters are incubated at 42 degrees C for about 48 hours.
* Positive results mean that microorganisms (bacteria etc) that are other than normal flora were found in the stool sample. Negative results mean that no abnormal microorganisms were detected in the sample.
Sensitivity
Broth dilution - This involves placing the organism in multiple plates or tubes before adding given concentrations of antibiotics.
Kirby-Bauer Method (disc diffusion) - Here, the antibiotic is placed in an agar plate that contains the organism and incubated for about 24 hours.
Microscopy
Microscopy techniques are used for identification of the parasitic organism responsible for the infection. For the sample to be observed under the microscope, then it has to be prepared.
Preparation may involve gram staining or preparing a wet mount. Through microscopy, it's possible to identify the microorganisms present in the sample by their morphological characteristics.
Learn more about Cell Culture and Tissue Culture
Return to studying Microorganisms
See Microscopy Imaging Techniques
Return to Microbiology as as Field of Study
Return from Microscopy Culture and Sensitivity to MicroscopeMaster home
References: Microscopy Culture and Sensitivity
Adugna Negussie et al. (2016). Bacteriological Profile and Antimicrobial Susceptibility Pattern of Blood Culture Isolates among Septicemia Suspected Children in Selected Hospitals Addis Ababa, Ethiopia.
Barbara H. Estridge, Anna P. Reynolds, and Norma J. Walters. (1990). Basic Medical Laboratory Techniques.
Loyola University Health System. (2017). Enhanced test for urinary tract infections detects more bacteria than standard test.
Susan M. Novak-Weekley and Michael Dunne, Jr. (2007). Blood Culturea key investigation for diagnosis of bloodstream infections.
Travis K. Price et al. (2016). The Clinical Urine Culture: Enhanced Techniques Improve Detection of Clinically Relevant Microorganisms.
Links
Find out how to advertise on MicroscopeMaster!