Endotoxin Vs Exotoxin
Definitions, Examples and Differences
Endotoxins - Definition
Discovered in the late 19th century, endotoxins are potentially toxic complex compounds commonly produced by Gram-negative bacteria. Also known as lipopolysaccharides (LPS), endotoxins are some of the most common pyrogens (substances with the potential to induce fever).
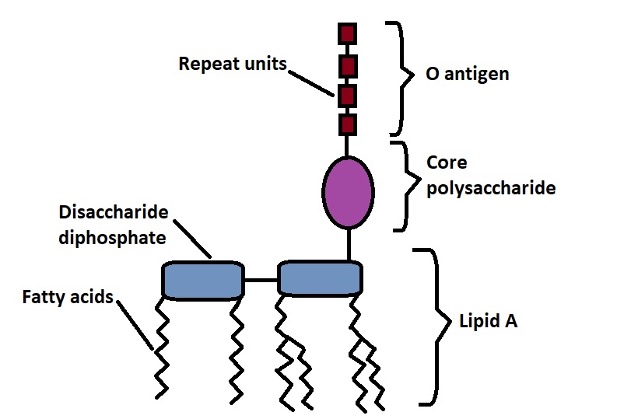
They comprise a hydrophilic polysaccharide and a hydrophobic component (Lipid A) and are part of the outer membrane of Gram-negative bacteria.
While endotoxins are components of the outer membrane in Gram-negative bacteria, they, like many other substances, are synthesized in the cytosol. From here, they are transported to the outer membrane through the inner membrane and the periplasmic space.
As mentioned, endotoxins consist of a polysaccharide linked to lipid A (a lipid moiety). Here, lipid A is responsible for the toxic effects of the compound.
Some of the symptoms associated with endotoxins include:
· High fever
· Vasodilation
· Diarrhea
· Shock
* The structure of endotoxins varies for different Gram-negative bacteria. For this reason, the level of toxicity is influenced by the structure of the endotoxin.
Endotoxin: Examples
The term endotoxin is generally used to refer to pyrogens produced by Gram-negative bacteria. In bacteriology, this complex compound is also known as lipopolysaccharide and can be found on the outer membranes of bacteria like Escherichia coli, Salmonella shigella, Vibrio cholerae, and Haemophilus influenzae.
Endotoxins are divided into two main categories that include:
Smooth lipopolysaccharides - Also referred to as wild-type LPS (sLPS), smooth lipopolysaccharide consists of the O-antigen, complete core oligosaccharides, as well as lipid A. Some of the bacterial species that contain smooth lipopolysaccharides on their surface are members of the genus Brucella. These include Brucella suis, Brucella, abortus, and Brucella melitensis.
Rough lipopolysaccharide - Unlike smooth lipopolysaccharide, rough lipopolysaccharide does not possess the O antigen. However, they still have lipid A and core oligosaccharides. In this case, oligosaccharides have been shown to be progressively shorter. Some of the organisms that contain rough lipopolysaccharides (also known as R species) include Brucella canis and Brucella ovis.
* While R species (bacteria species with rough lipopolysaccharide) also possess lipid A, studies have shown some of these species (e.g. Brucella) to be less virulent compared to those that possess rough lipopolysaccharide.
Characteristics of Endotoxins (Lipopolysaccharide)
As mentioned, lipopolysaccharides are commonly found on the outer membrane of most Gram-negative bacteria. However, due to structural differences, some of these endotoxins are not virulent and some of the species are not pathogenic.
While the toxicity of the compound is associated with the lipid A component, immunogenicity (the capacity to trigger an immune response) is due to the polysaccharide region.
While Gram-negative bacteria can release small amounts of the lipopolysaccharide, the endotoxin is generally less potent when compared to exotoxins. Because of their nature, they have been associated with a number of important functions including contributing to the permeability of the outer membrane.
Here, lipopolysaccharides enhance the permeability barrier, only allowing hydrophilic molecules of low molecular weight to pass through. On the other hand, they can promote adhesion (through the O antigens) of the organism to host cells thus contributing to the infection.
Exotoxins
Exotoxins may be described as toxins that are secreted by both Gram-negative and Gram-positive bacteria. They are highly potent and can cause severe damage to host cells by interfering with normal cellular processes among other mechanisms. Depending on the bacteria, exotoxins can be protein or non-protein.
Like many other molecules, these toxins are synthesized in the bacterial cytosol (e.g. botulinum neurotoxins) before they are released into the extracellular environment. As well, some of these bacteria can be found inside the host cells(as intracellular pathogens. For this reason, the toxins are directly released into the host cells.
While many bacteria secrete and release exotoxins, some only release these toxins following the lysis of the cell. Compared to endotoxins, exotoxins display significant variation in their molecular structure. Therefore, there are many types of exotoxins with many others yet to be described.
* Most exotoxins are proteins - They are produced during exponential growth.
* Exotoxins are diffusible and soluble proteins.
Types of Exotoxins
Type I Toxins
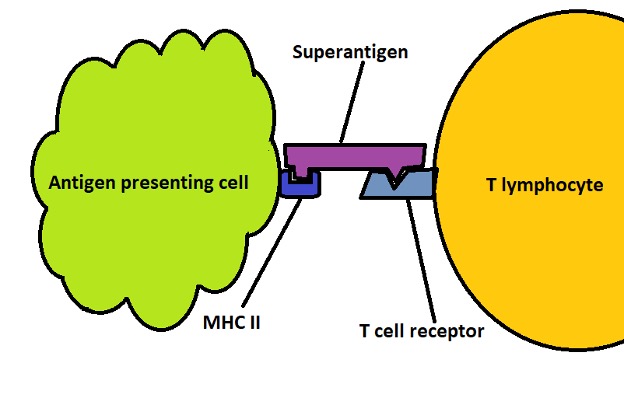
Also known as superantigens, type I toxins are membrane acting toxins that bind to the surface receptors of host cells thus stimulating transmembrane signals.
Some examples of type I toxins include:
· Streptococcal pyrogenic exotoxins
· Toxic shock syndrome toxin-1
· Enterotoxogenic E. coli enterotoxin
· Streptococcal superantigen
· Streptococcal mitogenic exotoxin
Compared to other antigens, type I toxins are capable of cross-linking the Major Histocompatibility Complex class II and T cell receptors which allows them to evade the antigen presenting stage of immune response. This also prevents the toxin from being engulfed and degraded.
By binding to both the Major Histocompatibility Complex class II molecules and T cell receptors, these toxins activate both types of cells (antigen-presenting cells and T cells) thus causing an overwhelming immune response.
Production of large amounts of T cells triggers the production of interleukin 2 (IL-2) and consequently the production of even more T cells. Some of the symptoms associated with high concentration of interleukin 2 in the bloodstream include diarrhea, high fever, and malaise, etc.
In some cases, increased secretion of interleukin 2 has been shown to trigger and influence the production of inflammatory cytokines like interleukin 1 and interleukin 8 among others. These cytokines can have a number of damaging effects including causing multiple organ failure, endothelial damage, and shock, etc.
Type II Toxins
Type II toxins are also known as membrane damaging toxins. As the name suggests, these exotoxins act by causing damage to the cell membrane.
Some examples of type II toxins include:
· Clostridium perfringens exotoxins - E.g. the alpha-toxin, the kappa toxin, and Epsilon toxin
· Leukotoxins
· Pseudomonas aeruginosa toxins - E.g. Phospholipase, Exotoxin U, Pyocyanin, and Cytotoxin
The different types of membrane damaging exotoxins use different mechanisms to disrupt the structural integrity of the cell membrane.
The following are some of the mechanisms through which these toxins cause damage to cells:
Alpha toxin - Produced by the bacterium Clostridium perfringens, Alpha toxin is a lecithinase that breaks down lecithin (a phospholipid) in the cell membrane. By breaking down this component, the toxin increases the permeability of capillary and muscle cells thereby contributing to gross edema.
When it enters the bloodstream, the toxin can be transported to different parts of the body causing damage to various organs.
Kappa toxin - Also produced by the bacterium Clostridium perfringens, Kappa toxin is a collagenase capable of breaking down connective tissue. Here, the enzyme breaks down the bonds in collagen thus affecting the integrity of extracellular structures.
By breaking down the connective tissue, the toxin causes mushy lesions of gas gangrene (myonecrosis). By causing damage to the connective tissue, the toxin also causes necrosis.
* By softening the affected tissue (by destroying the connective tissue), the toxin is able to continue spreading and causing further damage.
Mu toxin - The Mu toxin is a hyaluronidase produced by different strains of C. perfringens. As a members of the Hyaluronidases family, the toxin breaks down hyaluronic acid to release glucosamine. In chondrocyte and some other cells, hyaluronic acid acts as a cement that holds the cells in tissue together.
By breaking down this component, the toxin contributes to hemolytic and necrotic effects.
* Other exotoxins like Leukotoxins cause damage by forming pores in the cell membrane. In doing so, these toxins cause cell lysis.
Type III Toxins
Also known as intracellular effector toxins, type III toxins are A-B toxins; characterized by the "A" and "B" regions.
Some examples of type III exotoxins include:
· Anthrax toxins
· Botulism exotoxin
· Shiga toxin
· Cholera exotoxin
Compared to type I and Type II toxins, type III toxins act by disrupting given cellular processes or inactivating certain molecules (e.g. proteins). As mentioned, these toxins comprise two main parts (A and B). The "A" part of the active region serves to inactivate given intracellular molecules or signaling pathways. The B component, on the other hand, binds the toxin to specific surface receptors on the host cells.
Like many other molecules, type III exotoxins are secreted in the bacteria. Under the right circumstances, these toxins access the host cell and bind through the B subunit (of the toxin). While some of the toxins are translocated across the membrane into the cytosol, others enter the cell through endocytosis. Here, the toxins are engulfed in a vesicle and transported into the cell.
For the toxins that are internalized through the endosomes, there are two main destinations namely, the lysosome (where they are degraded) and the Golgi apparatus/endoplasmic reticulum. Toxins that end up in the lysosome undergo degradation while those that are transported to the Golgi apparatus or endoplasmic reticulum translocate to the cytosol.
In this case, it's the A subunit that translocates to the cytosol. Here, the toxin will act in a manner that negatively affects the cell.
Cholera Toxin (Enterotoxin)
One of the best examples of A-B toxin is the cholera toxin. Like some of the other bacterial A-B toxins, the exotoxin consist of 5 copies of the B subunit and a single copy of the A subunit. Using the B subunit, the toxin adheres to the ganglioside GM1 on the surface of intestinal cell membrane which allows it to remain attached to these cells (epithelial cells of the small intestine).
Adhesion of the toxin results in endocytosis with the toxin being transported to the endoplasmic reticulum.
Here, the A1 peptide of the A subunit is released to the A2 peptide through a reduction reaction allowing this peptide (A1) to translocate to the cytoplasm. Then, the peptide moves towards and binds to the G protein. G protein influences the actions of the enzyme adenylate cyclase located on the membrane.
By binding to the G protein, the A1 peptide prevents the G protein from cleaving GTP to GPD. As a result, the enzyme adenylate cyclase remains activated for a long period of time (abnormally) which results in increased levels of cAMP (cyclic AMP) in the cytosol.
High levels of cAMP activate the secretion of the enzyme Kinase A which in turn activates CFTR (cystic fibrosis transmembrane conductase regulator) allowing chloride ions to leave the cell.
This, in turn, influences water to leave the cell which causes diarrhea. This is a good example of how these exotoxins can affect given biological pathways thus causing disease.
Endotoxin Vs Exotoxin - Differences
Both endotoxins and exotoxins are produced by bacteria.
However, there are several differences between the two which include:
Type of bacteria - One of the main differences between endotoxins and exotoxins is with regard to the type of bacteria that secrete them. As mentioned, endotoxins are produced by Gram-negative bacteria. These are bacteria that have a thin cell wall and outer membrane (e.g. Escherichia coli and Salmonella enterica).
Exotoxins, on the other hand, are produced by both Gram-positive and Gram-negative bacteria. For this reason, there are many varieties of exotoxins with different modes of action. Clostridium botulinum is an example of Gram-positive bacteria that produces exotoxin while Vibrio cholerae is an example of Gram-negative bacteria that can produce exotoxin.
Location - While endotoxins are synthesized in the cytosol, they are translocated to the outer membrane of Gram-negative bacteria where they become components of the membrane. Here, they have also been associated with a number of functions including regulating the movement of material into the cell.
Exotoxins, on the other hand, are secreted in the cytosol and released into the external environment allowing them to interact with host cells. However, some exotoxins have been shown to remain in the cytosol until the cell undergoes lysis.
Heat stability - Endotoxins are described as being heat stable while exotoxins are typically heat labile. With the exception of Staphylococcal enterotoxin, most exotoxins are easily destroyed at about 60 degrees C. As well, endotoxins, which are heat stable, can survive in boiling water for about 30 minutes.
In other studies, they have been shown to survive and remain active in temperatures of up 250 degrees C. This makes endotoxins important components of the outer membrane in Gram-negative bacteria.
Genes - For the most part, genes involved in the production of exotoxins can be found in plasmid or bacteriophage (extrachromosomal DNA). Those involved in the production of endotoxins are typically found in the bacterial chromosome.
Toxicity - Compared to endotoxins, exotoxins can be released into the extracellular environment which allows them to interact with different types of cells. As such, they are more potent and exhibit higher toxicity when compared to endotoxins.
Solubility - Compared to endotoxins, exotoxins, which are diffusible proteins, are more soluble. Based on a number of studies, the solubility of endotoxins has been shown to vary significantly between endotoxins produced by different types of bacteria.
Some of the other differences between the two types of toxins include:
· Exotoxins are mostly polypeptides while endotoxins are lipopolysaccharide compounds
· Exotoxins are highly antigenic when compared to endotoxins
· Exotoxins exhibit higher receptor specificity compared to endotoxins
· Exotoxins exhibit enzymatic activity while endotoxins do not
· Toxoid can be formed from exotoxins but not from endotoxins - The term toxoid is used to refer to inactivated toxins (suppressed toxicity). Compared to exotoxins, these toxins are altered (not produced naturally by the bacteria) are generally used as vaccines. This is because they can induce an immune response as exotoxins without causing harm.
Bacteriology as a field of study
Bacterial Transformation, Conjugation
How do Bacteria cause Disease?
Bacteria - Size, Shape and Arrangement - Eubacteria
Return from Endotoxin Vs Exotoxin to MicroscopeMaster home
References
John D. Fraser. (2011). Clarifying the Mechanism of Superantigen Toxicity.
Lalita Mazgaeen and Prajwal Gurung. (2020). Recent Advances in Lipopolysaccharide Recognition Systems.
Stefan H. Gerber and Thomas C. Südhof. (2002). Molecular Determinants of Regulated Exocytosis.
Xiaoyuan Wang and Peter J. Quinn. (2016). Endotoxins: Structure, Function and Recognition.
Links
Find out how to advertise on MicroscopeMaster!